Drug Standards
- (2)
- (4)
- (3)
- (5)
- (665)
- (3)
- (1)
- (6)
- (27)
- (24)
- (1)
- (2)
- (35)
- (18)
- (1)
- (3)
- (3)
- (2)
- (2)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (2)
- (1)
- (2)
- (1)
- (2)
- (2)
- (1)
- (1)
- (1)
- (1)
- (1)
- (2)
- (1)
- (1)
- (1)
- (1)
- (1)
- (2)
- (1)
- (1)
- (2)
- (1)
- (1)
- (2)
- (2)
- (2)
- (1)
- (1)
- (2)
- (2)
- (1)
- (1)
- (3)
- (1)
- (1)
- (2)
- (3)
- (1)
- (1)
- (1)
- (2)
- (1)
- (3)
- (1)
- (1)
- (1)
- (2)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (2)
- (2)
- (2)
- (2)
- (1)
- (2)
- (2)
- (2)
- (1)
- (2)
- (2)
- (1)
- (1)
- (2)
- (2)
- (2)
- (2)
- (2)
- (1)
- (2)
- (2)
- (2)
- (1)
- (2)
- (2)
- (5)
- (1)
- (2)
- (2)
- (2)
- (2)
- (2)
- (1)
- (1)
- (1)
- (2)
- (2)
- (1)
- (1)
- (2)
- (2)
- (1)
- (2)
- (2)
- (1)
- (3)
- (1)
- (2)
- (2)
- (2)
- (2)
- (2)
- (5)
- (2)
- (2)
- (2)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (2)
- (1)
- (1)
- (1)
- (1)
- (2)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (1)
- (2)
- (1)
- (2)
- (1)
- (1)
- (1)
- (12)
- (38)
- (5)
- (1)
- (2)
- (1)
- (1)
- (12)
- (2)
- (15)
- (1)
- (1)
- (2)
- (1)
- (1)
- (1)
- (1)
- (1)
- (6)
- (15)
- (3)
- (1)
- (19)
- (3)
- (18)
- (1)
- (1)
- (1)
- (1)
- (2)
- (1)
- (29)
- (3)
- (2)
- (1)
- (2)
- (1)
- (1)
- (6)
- (1)
- (1)
- (1)
- (1)
- (44)
- (2)
- (29)
- (1)
- (73)
- (6)
- (73)
- (1)
- (1)
- (5)
- (1)
- (1)
- (2)
- (20)
- (4)
- (1)
- (1)
- (7)
- (8)
- (7)
- (5)
- (3)
- (5)
- (11)
- (14)
- (3)
- (1)
- (1)
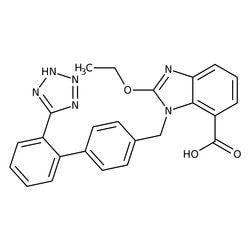
Filtered Search Results
Glycerin, Pharmaceutical Secondary Standard, Certified Reference Material, MilliporeSigma™ Supelco™
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Ibuprofen Impurity N Pharmaceutical Secondary Standard, MilliporeSigma™ Supelco™
Pharmaceutical secondary standards for application in quality control provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards
Ethylbenzene Pharmaceutical Secondary Standard, MilliporeSigma™ Supelco™
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards..
Theophylline Pharmaceutical Secondary Standard, MilliporeSigma™ Supelco™
Certified pharmaceutical secondary standards for application in quality control provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to pharmacopeia primary standards
Butylated hydroxyanisole Pharmaceutical Secondary Standard, MilliporeSigma™ Supelco™
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Guaifenesin Impurity B Pharmaceutical Secondary Standard, MilliporeSigma™ Supelco™
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
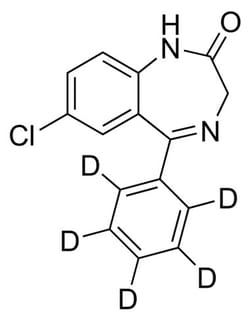
CERILLIANT™ Nordiazepam-d5 Solution, 1 mg/mL in methanol, Sold by MilliporeSigma™ Supelco™
This stable-labeled internal standard is suited for quantitation of nordiazepam levels in urine, serum, or plasma by LC/MS or GC/MS for urine drug testing, clinical toxicology, forensic analysis or isotope dilution methods.
Alfuzosin hydrochloride Pharmaceutical Secondary Standard, MilliporeSigma™ Supelco™
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Raloxifene Related Compound C Pharmaceutical Secondary Standard, MilliporeSigma™ Supelco™
Pharmaceutical secondary standards for application in quality control provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards