Organic zwitterions
- (1)
- (2)
- (37)
- (1)
- (15)
- (2)
- (27)
- (5)
- (2)
- (3)
- (2)
- (6)
- (2)
- (1)
- (59)
- (3)
- (1)
- (10)
- (1)
- (9)
- (1)
- (56)
- (1)
- (2)
- (14)
- (23)
- (3)
- (1)
- (3)
- (1)
- (1)
- (2)
- (4)
- (1)
- (2)
- (1)
- (2)
- (6)
- (3)
- (5)
- (14)
- (1)
- (4)
- (11)
- (8)
- (3)
- (9)
- (16)
- (1)
- (5)
- (9)
- (2)
- (6)
- (2)
- (15)
- (4)
- (7)
- (2)
- (1)
- (2)
- (5)
- (1)
- (1)
- (4)
- (4)
- (2)
- (1)
- (4)
- (1)
- (3)
- (10)
- (2)
- (2)
- (2)
- (2)
- (4)
- (2)
- (1)
- (2)
- (1)
- (5)
- (2)
- (1)
- (1)
- (1)
- (2)
- (2)
- (3)
- (2)
- (1)
- (1)
- (1)
- (4)
- (1)
- (8)
- (6)
- (4)
- (2)
- (1)
- (3)
- (4)
- (2)
- (2)
- (2)
- (3)
- (5)
- (3)
- (1)
- (2)
- (1)
- (1)
- (5)
- (3)
- (1)
- (2)
- (2)
- (2)
- (1)
- (1)
- (1)
- (2)
- (2)
- (7)
- (37)
- (1)
- (2)
- (4)
- (2)
- (1)
- (3)
- (8)
- (1)
- (3)
- (3)
- (3)
- (2)
- (20)
- (37)
- (62)
- (4)
- (9)
- (2)
- (4)
- (5)
- (3)
- (6)
- (2)
- (2)
- (2)
- (4)
- (6)
- (77)
- (3)
- (1)
- (1)
- (4)
- (45)
- (2)
- (1)
- (14)
- (2)
- (3)
- (1)
- (2)
- (3)
- (3)
- (3)
- (1)
- (3)
- (2)
- (2)
- (5)
- (4)
- (2)
- (2)
- (3)
- (2)
- (3)
- (2)
- (1)
- (3)
- (2)
- (12)
- (1)
- (4)
- (1)
- (3)
- (3)
- (4)
- (3)
- (2)
- (3)
- (1)
- (1)
- (2)
- (2)
- (2)
- (3)
Filtered Search Results
N-n-Hexyl-4-nitrobenzamide, 97%, Thermo Scientific™
CAS: 89399-21-3 Molecular Formula: C13H18N2O3 Molecular Weight (g/mol): 250.30 MDL Number: MFCD00229049 InChI Key: YIQCYVHPIUGJKJ-UHFFFAOYSA-N Synonym: benzamide, n-hexyl-4-nitro,n-n-hexyl-4-nitrobenzamide,n-hexyl-4-nitro-benzamide,acmc-20llm0,n-hexyl-p-nitrobenzamide PubChem CID: 4080064 IUPAC Name: N-hexyl-4-nitrobenzamide SMILES: CCCCCCNC(=O)C1=CC=C(C=C1)[N+]([O-])=O
| PubChem CID | 4080064 |
|---|---|
| CAS | 89399-21-3 |
| Molecular Weight (g/mol) | 250.30 |
| MDL Number | MFCD00229049 |
| SMILES | CCCCCCNC(=O)C1=CC=C(C=C1)[N+]([O-])=O |
| Synonym | benzamide, n-hexyl-4-nitro,n-n-hexyl-4-nitrobenzamide,n-hexyl-4-nitro-benzamide,acmc-20llm0,n-hexyl-p-nitrobenzamide |
| IUPAC Name | N-hexyl-4-nitrobenzamide |
| InChI Key | YIQCYVHPIUGJKJ-UHFFFAOYSA-N |
| Molecular Formula | C13H18N2O3 |
2,3,4-Trifluoro-6-nitroaniline, 98%, Thermo Scientific™
CAS: 148416-38-0 Molecular Formula: C6H3F3N2O2 Molecular Weight (g/mol): 192.097 MDL Number: MFCD00174089 InChI Key: ILENVKAVEFKZSD-UHFFFAOYSA-N Synonym: 6-nitro-2,3,4-trifluoroaniline,2-nitro-4,5,6-trifluoroaniline,pubchem4504,acmc-1bzfn,intermediates-zcf02138,ksc525q0b,attercop-chm at116509,buttpark 24\01-62,2,3,4-trifluoro-6-nitro-aniline,2,3,4-trifluoro-6-nitrophenylamine PubChem CID: 2775763 IUPAC Name: 2,3,4-trifluoro-6-nitroaniline SMILES: C1=C(C(=C(C(=C1F)F)F)N)[N+](=O)[O-]
| PubChem CID | 2775763 |
|---|---|
| CAS | 148416-38-0 |
| Molecular Weight (g/mol) | 192.097 |
| MDL Number | MFCD00174089 |
| SMILES | C1=C(C(=C(C(=C1F)F)F)N)[N+](=O)[O-] |
| Synonym | 6-nitro-2,3,4-trifluoroaniline,2-nitro-4,5,6-trifluoroaniline,pubchem4504,acmc-1bzfn,intermediates-zcf02138,ksc525q0b,attercop-chm at116509,buttpark 24\01-62,2,3,4-trifluoro-6-nitro-aniline,2,3,4-trifluoro-6-nitrophenylamine |
| IUPAC Name | 2,3,4-trifluoro-6-nitroaniline |
| InChI Key | ILENVKAVEFKZSD-UHFFFAOYSA-N |
| Molecular Formula | C6H3F3N2O2 |
N-(2-Ethylhexyl)-4-nitrobenzamide, 97%, Thermo Scientific™
CAS: 154702-08-6 Molecular Formula: C15H22N2O3 Molecular Weight (g/mol): 278.352 MDL Number: MFCD00709791 InChI Key: XYLYSNQGXATQBX-UHFFFAOYSA-N Synonym: n-2-ethylhexyl-4-nitrobenzamide PubChem CID: 4139944 IUPAC Name: N-(2-ethylhexyl)-4-nitrobenzamide SMILES: CCCCC(CC)CNC(=O)C1=CC=C(C=C1)[N+](=O)[O-]
| PubChem CID | 4139944 |
|---|---|
| CAS | 154702-08-6 |
| Molecular Weight (g/mol) | 278.352 |
| MDL Number | MFCD00709791 |
| SMILES | CCCCC(CC)CNC(=O)C1=CC=C(C=C1)[N+](=O)[O-] |
| Synonym | n-2-ethylhexyl-4-nitrobenzamide |
| IUPAC Name | N-(2-ethylhexyl)-4-nitrobenzamide |
| InChI Key | XYLYSNQGXATQBX-UHFFFAOYSA-N |
| Molecular Formula | C15H22N2O3 |
p-Nitrophenethyl alcohol, 98.1%, MP Biomedicals™
CAS: 100-27-6 Molecular Formula: C8H9NO3 Molecular Weight (g/mol): 167.164 InChI Key: IKMXRUOZUUKSON-UHFFFAOYSA-N Synonym: 4-nitrophenethyl alcohol,2-4-nitrophenyl ethanol,4-nitrobenzeneethanol,2-p-nitrophenyl ethanol,benzeneethanol, 4-nitro,p-nitrophenethyl alcohol,2-4-nitrophenyl ethan-1-ol,phenethyl alcohol, p-nitro,ccris 6079,4-nitrophenethylalcohol PubChem CID: 7494 IUPAC Name: 2-(4-nitrophenyl)ethanol SMILES: C1=CC(=CC=C1CCO)[N+](=O)[O-]
| PubChem CID | 7494 |
|---|---|
| CAS | 100-27-6 |
| Molecular Weight (g/mol) | 167.164 |
| SMILES | C1=CC(=CC=C1CCO)[N+](=O)[O-] |
| Synonym | 4-nitrophenethyl alcohol,2-4-nitrophenyl ethanol,4-nitrobenzeneethanol,2-p-nitrophenyl ethanol,benzeneethanol, 4-nitro,p-nitrophenethyl alcohol,2-4-nitrophenyl ethan-1-ol,phenethyl alcohol, p-nitro,ccris 6079,4-nitrophenethylalcohol |
| IUPAC Name | 2-(4-nitrophenyl)ethanol |
| InChI Key | IKMXRUOZUUKSON-UHFFFAOYSA-N |
| Molecular Formula | C8H9NO3 |
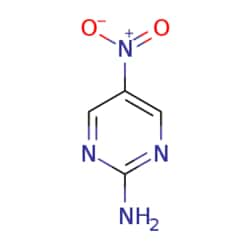
eMolecules 2-Amino-5-nitropyrimidine | 3073-77-6 | 1MG | Purity: 99%
Matrix Scientific | 2-Amino-5-nitropyrimidine | 1MG | 3073-77-6 | MFCD00006103

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Cayman Chemical Arachidonoyl p-Nitroaniline | 119520-58-0 | 424.6 g/mol |
Arachidonoyl p-Nitroaniline | 119520-58-0 | 424.6 g/mol |

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Cayman Chemical GW 9662, 22978-25-2, 10 mg
Molecular Formula C13H9ClN2O3, Purity ≥98%, Formula Weight 276.7, Synonyms: 2-chloro-5-nitrobenzanilide; The peroxisome proliferator-activated receptor (PPAR) is the nuclear receptor responsible for transducing the therapeutic activity of the thiazolidinediones. Thiazolidinediones are a group of structurally related synthetic PPAR agonists with antidiabetic actions in vivo. Rosiglitazone (BRL 49653) is a prototypical thiazolidinedione and has served as a reference compound for this class. There are many PPAR agonists, including 15-deoxy-.DELTA.12,14-prostaglandin J2 and azelaoyl PAF, which are naturally derived. However, only a few antagonists have been reported. GW 9662 blocks the PPAR-induced differentiation of monocytes to osteoclasts by >90% at a dose of 0.1 M. It is therefore a much more potent antagonist than BADGE, which is another reported PPAR antagonist.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More