Learn More
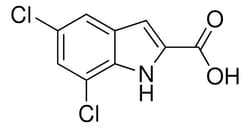
Sigma Aldrich 5,7-Dichloro-1H-indole-2-carboxylic acid
Shop All MilliporeSigma Sigma Organic Chemistry Products

Description
- 3-butyn-2-one undergoes asymmetric double-michael reaction with ortho-tosylamidophenyl malonate catalyzed by chiral aminophosphines to yield indolines
- It undergoes double michael reaction with nitrogen-containing tethered diacid to give pipecolic acid derivatives.3-butyn-2-one was used in the synthesis of clerodane diterpenoid (+/-)-sacacarin
- It was used as substrate in stereoselective, conjugate arylation mediated by gallium(iii) chloride leading to (e)-α,α-unsaturated ketones
Specifications
Specifications
| Molecular Formula | C9H5Cl2NO2 |
| Linear Formula | C9H5Cl2NO2 |
| MDL Number | MFCD02664420 |
| Quantity | 2.5 mL |
| Molecular Weight (g/mol) | 230.05 |
By clicking Submit, you acknowledge that you may be contacted by Fisher Scientific in regards to the feedback you have provided in this form. We will not share your information for any other purposes. All contact information provided shall also be maintained in accordance with our Privacy Policy.