Learn More
Description
Includes
- bFGF Microtiter plate, 96 wells
- Biotin Conjugate, 7mL
- Avidin Conjugate, 14mL
- bFGF Standard, 2 vials
- Calibrator diluent I, 25mL
- Calibrator diluent II, 25mL
- Wash Buffer (20x), 60mL
- Substrate A, 10mL
- Substrate B, 10mL
- Stop solution, 14mL
Specifications
Specifications
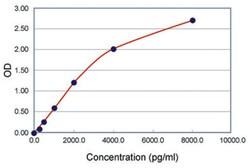
| Calibration Range | 78 to 8000pg/mL |
| Conjugate | HRP |
| Cross Reactivity | Human |
| Description | FGF2 (Human) ELISA Kit is a sandwich enzyme immunoassay for the quantitative measurement of human FGF2. |
| Detection Limit | 78pg/mL |
| Detection Method | Colorimetric |
| Quantity | 1 Kit |
| Regulatory Status | For research use only (RUO) |
| Sample Type | Cell Culture Supernatant, Plasma, Serum |
| Sample Volume | 100 μL |
| Show More |
By clicking Submit, you acknowledge that you may be contacted by Fisher Scientific in regards to the feedback you have provided in this form. We will not share your information for any other purposes. All contact information provided shall also be maintained in accordance with our Privacy Policy.