Learn More
Specifications
Specifications
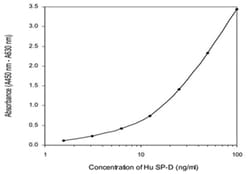
| Calibration Range | 1.56 to 100ng/mL |
| Conjugate | HRP |
| Cross Reactivity | Bovine, Horse, Human, Monkey |
| Description | SFTPD (Human) ELISA Kit is a sandwich enzyme immunoassay for the quantitative measurement of human SFTPD in serum, plasma (-EDTA, -Heparin, -Citrate), amniotic fluid, bronchoalveolar lavage. |
| Detection Limit | 0.01ng/mL |
| Detection Method | Colorimetric |
| Includes | 96-well Antibody coated microtiter strip, 13mL Biotin labelled Antibody,13mL Streptavidin HRP Conjugate, 1 vial Master standards, 2 vials Quality control high, 2 vials Quality control low, 50mL Dilution Buffer, 100mL Conc. (10X) Wash solution, 13mL Substrate solution, 13mL stop solution |
| Quantity | 1 Kit |
| Sample Type | Amniotic Fluid, Bronchoalveolar Lavage fluid, Plasma (Citrate, EDTA, Heparin), Serum |
| Sample Volume | 10 μL |
| Show More |
By clicking Submit, you acknowledge that you may be contacted by Fisher Scientific in regards to the feedback you have provided in this form. We will not share your information for any other purposes. All contact information provided shall also be maintained in accordance with our Privacy Policy.