Learn More
Sigma Aldrich Lenalidomide-Photoswitch2-Nh2 Hydrochloride
Shop All MilliporeSigma Sigma Organic Chemistry Products
Description
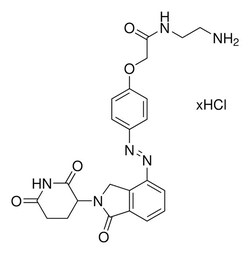
Protein degrader building block Lenalidomide-Photoswitch2-NH2 hydrochloride enables the synthesis of PHOtochemically TArgeting Chimeras (PHOTACs), or photoswitchable proteolysis-targeting chimeras (PROTACs) that can be reversibly activated with different wavelengths of light. Developed in the Trauner and Pagano labs, PHOTACs are inactive in the dark but are activated to the cis isomer via irradiation at 390 nm and reversibly deactivated at wavelengths above 450 nm. As described in Reynders et al, this conjugate was used to prepare PHOTAC-II-5 and contains a Cereblon (CRBN)-recruiting ligand, an azobenzene photoswitchable crosslinker, and a pendant amine for reactivity with an acid on the target warhead. Light-mediated control of the resulting PHOTAC affords advanced temporal and spatial control of targeted protein degradation. Suggested wavelengths for photoswitching:
- Switch to cis isomer: 390 nm (380-400 nm)
- Switch to trans isomer (thermally more stable isomer): >450 nm. Low-intensity light needed for photoactivation is not cytotoxicBrowse our full offering of degrader building blocks that streamlines the synthesis of degrader libraries.
Technology Spotlight: Degrader Building Blocks for Targeted Protein Degradation. Portal: Building PROTAC™ Degraders for Targeted Protein Degradation. PHOTACs enable optical control of protein degradationPhotoPROTACs: A Novel Biotechnology for Cancer TreatmentPROTAC is a registered trademark of Arvinas Operations, Inc., and is used under license
Specifications
Specifications
| CAS | 632496-52-6 |
| Quantity | 250 mg |
By clicking Submit, you acknowledge that you may be contacted by Fisher Scientific in regards to the feedback you have provided in this form. We will not share your information for any other purposes. All contact information provided shall also be maintained in accordance with our Privacy Policy.