Learn More
Invitrogen™ Mouse TRANCE (RANKL) Recombinant Protein
Description
Recombinant mouse RANK Ligand is a non-glycosylated protein containing 175 amino acids. Reconstitute using sterile water at 0.1 mg/mL and centrifuge prior to opening vial. Gently pipet solution down the sides of the vial. DO NOT VORTEX sample. Store reconstituted material at -20°C and add 0.1% BSA for additional stability.

Specifications
Specifications
| Accession Number | O35235 |
| Concentration | 0.1 mg/mL |
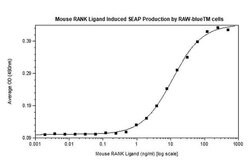
| For Use With (Application) | Control, Bioactivity |
| Formulation | PBS with no preservative; pH 7.5 |
| Gene ID (Entrez) | 21943 |
| Molecular Weight (g/mol) | 19.5 kDa |
| Name | Mouse TRANCE (RANKL) |
| pH Range | 7.5 |
| Purification Method | Purified |
| Quantity | 10 μg |
| Show More |
Frequently Asked Questions (FAQs)
There is no direct correlation or calculation between specific activity unit and International Unit (IU) values. International Units (IU) express a quantification of activity for the base amount of a substance in relation to an analogous reference standard with an internationally accepted unit of biological potency (i.e., IU/ng) that has been assigned based on an International Collaborative Study conducted by the World Health Organization (WHO). WHO Reference Standards are made available by the National Institute for Biological Standards and Control (NIBSC). Intended to simplify the comparison of activity of a substance obtained from different sources, IU measurements can vary as comparison methods are rarely the same between sources. A true direct comparison requires standardized methods of analysis in order to guarantee comparability of the substance’s activity in relation to its mass across sources.
Gibco recombinant proteins are frequently formulated without carrier proteins or additives (e.g., BSA, HSA, sucrose, etc.) and Gibco PeproTech recombinant proteins don't contain a carrier protein. As a result, during lyophilization, the protein product may be deposited on the vial as a thin, and sometimes invisible film instead of a pellet. The size of the pellet, if any, is not directly related to the quantity of the recombinant protein in the vial. Our quality control procedures assure that each vial contains the correct amount of product.
To ensure complete recovery of protein product, before opening a vial of lyophilized recombinant protein, we recommend centrifuging it in a microcentrifuge for 20-30 seconds to drive any protein that may be lodged in the cap or on the side or the bottom of the vial. After reconstitution, you can confirm the presence of product protein by running a small amount on SDS-PAGE. In general, a protein band with expected size should be visible with as little as 10 ng of protein loaded on an acrylamide gel.
Protein sequence information is available on the individual product page, except for proprietary engineered proteins.
In general, we recommend using polypropylene tubes for storing aliquots of reconstituted recombinant proteins. Specific information for appropriate storage and handling of many recombinant proteins can be found on the product pages.
No, each lot can vary slightly with regards to its specific activity. The information on the data sheet for each product is provided as a guide. For lot-specific information, please please check the Certificate of Analysis or contact Technical Support.
By clicking Submit, you acknowledge that you may be contacted by Fisher Scientific in regards to the feedback you have provided in this form. We will not share your information for any other purposes. All contact information provided shall also be maintained in accordance with our Privacy Policy.