Learn More
R&D Systems™ Recombinant SARS-CoV-2 Y453F Spike RBD Fc Chimera Protein CF
Description
Specifications
Specifications
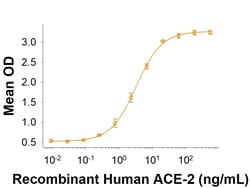
| For Use With (Application) | Bioactivity |
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. |
| Molecular Weight (g/mol) | M.W.-observed: 57-64 kDa, under reducing conditions., M.W.-theroretical: 52 kDa |
| Quantity | 100 μg |
| Source | Human embryonic kidney cell, HEK293-derived sars-cov-2 Spike RBD protein SARS-CoV-2 Spike RBD (Arg319-Phe541)(Tyr453Phe) (N-terminus) Accession # YP_009724390.1 IEGRMD Human IgG1 (Pro100-Lys330) (C-terminus) |
| Storage Requirements | Use a manual defrost freezer and avoid repeated freeze-thaw cycles. 12 months from date of receipt, -20°C to -70°C as supplied. 1 month, 2°C to 8°C under sterile conditions after reconstitution. 3 months, -20°C to -70°C under sterile conditions after reconstitution. |
| Endotoxin Concentration | <0.10 EU / 1 μg of the protein by the LAL method. |
| Conjugate | Unconjugated |
| Reconstitution | Reconstitute at 500 μg/mL in PBS. |
| Purity or Quality Grade | >95%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie™ Blue Staining. |
| Show More |
By clicking Submit, you acknowledge that you may be contacted by Fisher Scientific in regards to the feedback you have provided in this form. We will not share your information for any other purposes. All contact information provided shall also be maintained in accordance with our Privacy Policy.