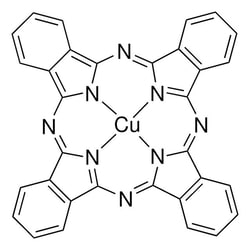
Tetrapyrroles and derivatives
- (2)
- (2)
- (5)
- (1)
- (2)
- (2)
- (6)
- (3)
- (1)
- (2)
- (2)
- (9)
- (2)
- (2)
- (2)
- (2)
Filtered Search Results
Copper(II) 5,9,14,18,23,27,32,36-Octabutoxy-2,3-naphthalocyanine 97.0+%, TCI America™
CAS: 155773-67-4 Molecular Formula: C80H88CuN8O8 Molecular Weight (g/mol): 1353.178 MDL Number: MFCD00191950 InChI Key: JJIWQZDKAOMTKU-UHFFFAOYSA-N Synonym: 5,9,14,18,23,27,32,36-Octabutoxy-2,3-naphthalocyanine Copper(II) PubChem CID: 22834032 SMILES: CCCCOC1=C2C=CC=CC2=C(C3=C4[N-]C(=C31)N=C5C6=C(C7=CC=CC=C7C(=C6C(=NC8=NC(=NC9=NC(=N4)C1=C(C2=CC=CC=C2C(=C19)OCCCC)OCCCC)C1=C(C2=CC=CC=C2C(=C18)OCCCC)OCCCC)[N-]5)OCCCC)OCCCC)OCCCC.[Cu+2]
| PubChem CID | 22834032 |
|---|---|
| CAS | 155773-67-4 |
| Molecular Weight (g/mol) | 1353.178 |
| MDL Number | MFCD00191950 |
| SMILES | CCCCOC1=C2C=CC=CC2=C(C3=C4[N-]C(=C31)N=C5C6=C(C7=CC=CC=C7C(=C6C(=NC8=NC(=NC9=NC(=N4)C1=C(C2=CC=CC=C2C(=C19)OCCCC)OCCCC)C1=C(C2=CC=CC=C2C(=C18)OCCCC)OCCCC)[N-]5)OCCCC)OCCCC)OCCCC.[Cu+2] |
| Synonym | 5,9,14,18,23,27,32,36-Octabutoxy-2,3-naphthalocyanine Copper(II) |
| InChI Key | JJIWQZDKAOMTKU-UHFFFAOYSA-N |
| Molecular Formula | C80H88CuN8O8 |
1,2,3,4,8,9,10,11,15,16,17,18,22,23,24,25-Hexadecafluorophthalocyanine Copper(II) (purified by sublimation) 98.0+%, TCI America™
CAS: 14916-87-1 Molecular Formula: C32H2CuF16N8 Molecular Weight (g/mol): 865.944 MDL Number: MFCD00274642 InChI Key: NKBVANSQKHVZHF-UHFFFAOYSA-N Synonym: Copper(II) 1,2,3,4,8,9,10,11,15,16,17,18,22,23,24,25-Hexadecafluorophthalocyanine, F16CuPc PubChem CID: 87077853 SMILES: C12=C(C(=C(C(=C1F)F)F)F)C3=NC4=NC(=NC5=NC(=NC6=C7C(=C(N6)N=C2N3)C(=C(C(=C7F)F)F)F)C8=C5C(=C(C(=C8F)F)F)F)C9=C4C(=C(C(=C9F)F)F)F.[Cu]
| PubChem CID | 87077853 |
|---|---|
| CAS | 14916-87-1 |
| Molecular Weight (g/mol) | 865.944 |
| MDL Number | MFCD00274642 |
| SMILES | C12=C(C(=C(C(=C1F)F)F)F)C3=NC4=NC(=NC5=NC(=NC6=C7C(=C(N6)N=C2N3)C(=C(C(=C7F)F)F)F)C8=C5C(=C(C(=C8F)F)F)F)C9=C4C(=C(C(=C9F)F)F)F.[Cu] |
| Synonym | Copper(II) 1,2,3,4,8,9,10,11,15,16,17,18,22,23,24,25-Hexadecafluorophthalocyanine, F16CuPc |
| InChI Key | NKBVANSQKHVZHF-UHFFFAOYSA-N |
| Molecular Formula | C32H2CuF16N8 |
TPP (=Tetraphenylporphyrin) [Ultra-high sensitive spectrophotometric reagent for Cu], TCI America™
CAS: 917-23-7 Molecular Formula: C44H30N4 Molecular Weight (g/mol): 614.75 MDL Number: MFCD00011680 InChI Key: AQPPOLXYUQPDOD-UHFFFAOYSA-N Synonym: tetraphenylporphyrin,5,10,15,20-tetraphenylporphyrin,meso-tetraphenylporphine,tetraphenylporphine,5,10,15,20-tetraphenylporphine,meso-tetraphenylporphyrin,21h,23h-porphine, 5,10,15,20-tetraphenyl,5,10,15,20-tetraphenyl-21h,23h-porphine,meso-tetraphenylporphyrine,tetraphenyl porphine PubChem CID: 70186 IUPAC Name: 2,7,12,17-tetraphenyl-21,22,23,24-tetraazapentacyclo[16.2.1.1³,⁶.1⁸,¹¹.1¹³,¹⁶]tetracosa-1,3,5,7,9,11,13(22),14,16,18(21),19-undecaene SMILES: N1C2=CC=C1C(=C1C=CC(=N1)C(=C1C=CC(=N1)C(=C1NC(C=C1)=C2C1=CC=CC=C1)C1=CC=CC=C1)C1=CC=CC=C1)C1=CC=CC=C1
| PubChem CID | 70186 |
|---|---|
| CAS | 917-23-7 |
| Molecular Weight (g/mol) | 614.75 |
| MDL Number | MFCD00011680 |
| SMILES | N1C2=CC=C1C(=C1C=CC(=N1)C(=C1C=CC(=N1)C(=C1NC(C=C1)=C2C1=CC=CC=C1)C1=CC=CC=C1)C1=CC=CC=C1)C1=CC=CC=C1 |
| Synonym | tetraphenylporphyrin,5,10,15,20-tetraphenylporphyrin,meso-tetraphenylporphine,tetraphenylporphine,5,10,15,20-tetraphenylporphine,meso-tetraphenylporphyrin,21h,23h-porphine, 5,10,15,20-tetraphenyl,5,10,15,20-tetraphenyl-21h,23h-porphine,meso-tetraphenylporphyrine,tetraphenyl porphine |
| IUPAC Name | 2,7,12,17-tetraphenyl-21,22,23,24-tetraazapentacyclo[16.2.1.1³,⁶.1⁸,¹¹.1¹³,¹⁶]tetracosa-1,3,5,7,9,11,13(22),14,16,18(21),19-undecaene |
| InChI Key | AQPPOLXYUQPDOD-UHFFFAOYSA-N |
| Molecular Formula | C44H30N4 |
Sigma Aldrich Copper(II) phthalocyanine
MilliporeSigma Sigma Organics products encompass a wide range of quality reagents, solvents, catalysts, and building blocks for organic synthesis. From benchtop discovery to process development and scale-up, Sigma Organics solutions are built to meet the needs of synthetic chemists.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
| Linear Formula | C32H16CuN8 |
|---|---|
| CAS | 147-14-8 |
| Molecular Weight (g/mol) | 576.07 |
| MDL Number | MFCD00010719 |
| Synonym | CuPc; Phthalocyanine blue; Pigment Blue 15 |
| RTECS Number | GL8510000 |
| Recommended Storage | Room Temperature |
| Molecular Formula | C32H16CuN8 |
| EINECS Number | 205-685-1 |
Sigma Aldrich Copper(II) phthalocyanine
MilliporeSigma Sigma Organics products encompass a wide range of quality reagents, solvents, catalysts, and building blocks for organic synthesis. From benchtop discovery to process development and scale-up, Sigma Organics solutions are built to meet the needs of synthetic chemists.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
| Linear Formula | C32H16CuN8 |
|---|---|
| CAS | 147-14-8 |
| Molecular Weight (g/mol) | 576.07 |
| MDL Number | MFCD00010719 |
| Synonym | CuPc; Phthalocyanine blue; Pigment Blue 15 |
| RTECS Number | GL8510000 |
| Recommended Storage | Room Temperature |
| Molecular Formula | C32H16CuN8 |
| EINECS Number | 205-685-1 |
Sigma Aldrich Copper(II) phthalocyanine
MilliporeSigma Sigma Organics products encompass a wide range of quality reagents, solvents, catalysts, and building blocks for organic synthesis. From benchtop discovery to process development and scale-up, Sigma Organics solutions are built to meet the needs of synthetic chemists.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
TCPP 97.0+%, TCI America™
CAS: 14609-54-2 Molecular Formula: C48H30N4O8 Molecular Weight (g/mol): 790.788 MDL Number: MFCD00064860 InChI Key: SMOZAZLNDSFWAB-UHFFFAOYSA-N Synonym: Tetrakis(4-carboxyphenyl)porphyrin PubChem CID: 5479495 IUPAC Name: 4-[10,15,20-tris(4-carboxyphenyl)-21,24-dihydroporphyrin-5-yl]benzoic acid SMILES: C1=CC(=CC=C1C2=C3C=CC(=C(C4=CC=C(N4)C(=C5C=CC(=N5)C(=C6C=CC2=N6)C7=CC=C(C=C7)C(=O)O)C8=CC=C(C=C8)C(=O)O)C9=CC=C(C=C9)C(=O)O)N3)C(=O)O
| PubChem CID | 5479495 |
|---|---|
| CAS | 14609-54-2 |
| Molecular Weight (g/mol) | 790.788 |
| MDL Number | MFCD00064860 |
| SMILES | C1=CC(=CC=C1C2=C3C=CC(=C(C4=CC=C(N4)C(=C5C=CC(=N5)C(=C6C=CC2=N6)C7=CC=C(C=C7)C(=O)O)C8=CC=C(C=C8)C(=O)O)C9=CC=C(C=C9)C(=O)O)N3)C(=O)O |
| Synonym | Tetrakis(4-carboxyphenyl)porphyrin |
| IUPAC Name | 4-[10,15,20-tris(4-carboxyphenyl)-21,24-dihydroporphyrin-5-yl]benzoic acid |
| InChI Key | SMOZAZLNDSFWAB-UHFFFAOYSA-N |
| Molecular Formula | C48H30N4O8 |
TMPyP 98.0+%, TCI America™
CAS: 36951-72-1 Molecular Formula: C72H66N8O12S4 Molecular Weight (g/mol): 1363.604 MDL Number: MFCD00013468 InChI Key: AKZFRMNXBLFDNN-UHFFFAOYSA-K Synonym: tmpyp,tmpyp4 tosylate,tmpyp tetratosylate,tmpyp4 tetratosylate,tppt 4,meso-tetra 4-n-methyl-pyridyl porphyrin,meso-tetrakis n-methyl-4-pyridyl porphine tetratosylate salt,meso-tetrakis n-methylpyridinium-4-yl porphine tetratosylate,meso-tetrakis n-methyl-4-pyridyl porphine tetrakis p-toluenesulfonate,5,10,15,20-tetrakis 1-methylpyridinium-4-yl porphyrin tetra p-toluenesulfonate PubChem CID: 11979833 IUPAC Name: 4-methylbenzenesulfonate;5,10,15,20-tetrakis(1-methylpyridin-1-ium-4-yl)-21,22-dihydroporphyrin SMILES: CC1=CC=C(C=C1)S(=O)(=O)[O-].CC1=CC=C(C=C1)S(=O)(=O)[O-].CC1=CC=C(C=C1)S(=O)(=O)[O-].CC1=CC=C(C=C1)S(=O)(=O)[O-].C[N+]1=CC=C(C=C1)C2=C3C=CC(=C(C4=CC=C(N4)C(=C5C=CC(=N5)C(=C6C=CC2=N6)C7=CC=[N+](C=C7)C)C8=CC=[N+](C=C8)C)C9=CC=[N+](C=C9)C)N3
| PubChem CID | 11979833 |
|---|---|
| CAS | 36951-72-1 |
| Molecular Weight (g/mol) | 1363.604 |
| MDL Number | MFCD00013468 |
| SMILES | CC1=CC=C(C=C1)S(=O)(=O)[O-].CC1=CC=C(C=C1)S(=O)(=O)[O-].CC1=CC=C(C=C1)S(=O)(=O)[O-].CC1=CC=C(C=C1)S(=O)(=O)[O-].C[N+]1=CC=C(C=C1)C2=C3C=CC(=C(C4=CC=C(N4)C(=C5C=CC(=N5)C(=C6C=CC2=N6)C7=CC=[N+](C=C7)C)C8=CC=[N+](C=C8)C)C9=CC=[N+](C=C9)C)N3 |
| Synonym | tmpyp,tmpyp4 tosylate,tmpyp tetratosylate,tmpyp4 tetratosylate,tppt 4,meso-tetra 4-n-methyl-pyridyl porphyrin,meso-tetrakis n-methyl-4-pyridyl porphine tetratosylate salt,meso-tetrakis n-methylpyridinium-4-yl porphine tetratosylate,meso-tetrakis n-methyl-4-pyridyl porphine tetrakis p-toluenesulfonate,5,10,15,20-tetrakis 1-methylpyridinium-4-yl porphyrin tetra p-toluenesulfonate |
| IUPAC Name | 4-methylbenzenesulfonate;5,10,15,20-tetrakis(1-methylpyridin-1-ium-4-yl)-21,22-dihydroporphyrin |
| InChI Key | AKZFRMNXBLFDNN-UHFFFAOYSA-K |
| Molecular Formula | C72H66N8O12S4 |
Medchemexpress LLC Bilirubin oxidase (BOD) | 80619-01-8 | ≥10 U/mg solid | 1000 U
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Bilirubin oxidase (BOD) is a multi-copper oxidase that catalyzes the oxidation of bilirubin to biliverdin and reduces molecular oxygen to water. It participates in the metabolism of porphyrin and chlorophyll. It is widely used in biochemical research as a catalyst for oxygen reduction.
- Catalyzes oxidation of bilirubin to biliverdin
- Reduces molecular oxygen to water
- Participates in metabolism of porphyrin and chlorophyll
- Used as a catalyst for oxygen reduction in biochemical research

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC Bilirubin Oxidase, Myrothecium verrucaria | 80619-01-8 | 250 U
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Bilirubin oxidase (BOD) is a multi-copper oxidase that catalyzes the oxidation of bilirubin to biliverdin, reducing molecular oxygen to water. Sourced from Myrothecium verrucaria, it plays a role in the metabolism of porphyrins and chlorophyll, making it valuable for biochemical research as an oxygen reduction catalyst.
- Catalyzes the oxidation of bilirubin to biliverdin
- Reduces molecular oxygen to water
- Involved in the metabolism of porphyrins and chlorophyll
- Acts on the CH-CH group as a donor, using oxygen as an acceptor
- Substrates include bilirubin and O2
- Products include biliverdin and H2O
- Used in biochemical research
- Appearance is a light blue to blue solid

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC Bilirubin oxidase | 80619-01-8 | 500 U
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Bilirubin oxidase (BOD) is a multi-copper oxidase that catalyzes the oxidation of bilirubin to biliverdin and reduces molecular oxygen to water. It participates in the metabolism of porphyrin and chlorophyll and is widely used in biochemical research as a catalyst for oxygen reduction.
- Catalyzes the oxidation of bilirubin to biliverdin
- Reduces molecular oxygen to water
- Participates in porphyrin and chlorophyll metabolism
- Functions as an oxygen reduction catalyst in biochemical research
- Appearance as a solid
- Color ranges from light blue to blue
- Intended for research use only
- Unit defined as the amount of enzyme converting 1 μmol of bilirubin into biliverdin per minute at 25 °C and pH 8.0

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC Pyraclonil | 158353-15-2 | 97.0% | 314.77 | 50 MG
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Pyraclonil is a chemical compound that acts as a protoporphyrinogen oxidase (PPO) inhibitor. It functions as a herbicide and demonstrates high effectiveness in controlling both susceptible and multiple-herbicide-resistant populations of *E. indica*. Additionally, Pyraclonil is noted for its role as a click chemistry reagent, containing an Alkyne group which allows it to undergo copper-catalyzed azide-alkyne cycloaddition (CuAAc) reactions with molecules possessing Azide groups.
- Mechanism of action: PPO inhibitor.
- Application: herbicide, effective against susceptible and multiple-herbicide-resistant *E. indica*.
- Chemical reactivity: click chemistry reagent with an alkyne group, capable of CuAAc reactions with azide-containing molecules.
- In vitro efficacy: shows good control in vitro, leading to 94.5-94.9% and 89.5-90.4% fresh-weight reductions in susceptible and resistant populations, respectively.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More


![TPP (=Tetraphenylporphyrin) [Ultra-high sensitive spectrophotometric reagent for Cu], TCI America™](https://assets.fishersci.com/TFS-Assets/CCG/Chemical-Structures/chemical-structure-cas-917-23-7.jpg-250.jpg)