Learn More
Description
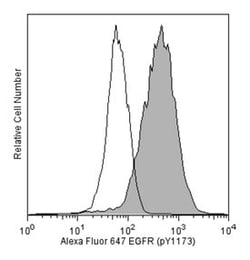
Epidermal Growth Factor (EGF) elicits a variety of cellular responses that are initiated by EGF Receptor (EGFR) binding and activation of intrinsic tyrosine kinase activity. EGFR, also known as ErbB1 or HER1, is a member of the ErbB class of receptor protein tyrosine kinases. It has an extracellular ligand-binding domain, a single transmembrane region, and a cytoplasmic region containing a protein tyrosine kinase domain and a c-terminal regulatory domain with many phosphorylation sites. Following ligand binding, EGFR forms homodimers and heterodimers with ErbB2. Specific C-terminal tyrosine residues are then autophosphorylated and, in turn, bind to adaptor proteins, kinases, or protein tyrosine phosphatases. Specifically, phosphorylated tyrosine 1173 (Y1173) interacts with Shc, SHP1, and PLCγ, which mediate downstream signaling cascades and negative feedback regulation of EGFR activation. Inappropriate expression or mutations of EGFR and/or deregulation of its signaling pathways are associated with many types of cancer, making EGFR a promising target for cancer therapies. The 9H2 monoclonal antibody recognizes the phosphorylated Y1173 in the regulatory domain of human EGFR.
Intracellular Staining

Specifications
Specifications
| Antigen | EGF Receptor (pY1173) |
| Applications | Flow Cytometry |
| Classification | Monoclonal |
| Clone | 9H2 |
| Conjugate | Alexa Fluor 647 |
| Formulation | Aqueous buffered solution containing BSA and ≤0.09% sodium azide. |
| Host Species | Mouse |
| Immunogen | Phosphorylated Human EGF Receptor Peptide |
| Purification Method | Affinity Purified |
| Quantity | 50 Tests |
| Show More |
By clicking Submit, you acknowledge that you may be contacted by Fisher Scientific in regards to the feedback you have provided in this form. We will not share your information for any other purposes. All contact information provided shall also be maintained in accordance with our Privacy Policy.