Learn More
Description
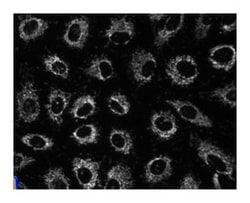
Heat shock proteins (Hsp) are a set of highly conserved proteins that include constitutively expressed (Hsp60, Hsp70, and Hsp90) and stress-induced (Hsp27 and Hsp72) proteins. Hsp60 is localized to the mitochondria, where it promotes mitochondrial protein folding and facilitates proteolytic degradation of misfolded or denatured proteins. It binds Hsp10, which regulates the substrate binding and ATPase activity of Hsp60. In HeLa and Jurkat mitochondria, Hsp60 associates with caspase-3 to form a complex that dissociates and releases from the mitochondria during apoptosis. In addition, Hsp60 accelerates the maturation of procaspase-3 through its ATP-dependent “foldase” activity. In addition to its role in protein folding, Hsp60 has also been implicated in immune function. In macrophages, its binding to the toll-like receptor-4 complex induces production of TNFα and nitric oxide and stimulation of a proinflammatory response. Thus, the protein folding function of Hsp60 is involved in mitochondrial protein folding in both normal and apoptotic cells, while release of Hsp60 during necrosis is thought to stimulate a proinflammatory response.
Host Species: Mouse
Clone: 24
Isotype: IgG1
Species Reactivity: Human
Immunogen: Human Hsp60 aa. 1-200
Formula Weight [Chemical]: 60kDa
Immunofluorescence, Western Blotting

Specifications
Specifications
| Antigen | Hsp60 |
| Applications | Western Blot |
| Classification | Monoclonal |
| Clone | 24/HSP60 |
| Concentration | 250μg/mL |
| Conjugate | Unconjugated |
| Formulation | Aqueous buffered solution containing BSA, glycerol, and ≤0.09% sodium azide. |
| Host Species | Mouse |
| Immunogen | Human Hsp60 aa. 1-200 |
| Purification Method | Affinity Purified |
| Show More |
Safety and Handling
For Research Use Only.
By clicking Submit, you acknowledge that you may be contacted by Fisher Scientific in regards to the feedback you have provided in this form. We will not share your information for any other purposes. All contact information provided shall also be maintained in accordance with our Privacy Policy.