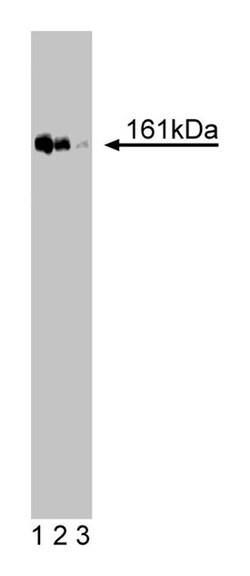Learn More
Description
Eukaryotic DNA topoisomerase II, a ubiquitous ATP-dependent type II topoisomerase, is an essential nuclear enzyme in DNA replication and transcription, chromatin segregation, and cell cycle progression. Topoisomerases transiently break a pair of complementary strands in double-stranded DNA to form a gate for the passage of duplex DNA. Two isoforms of DNA topoisomerase II have been identified: topo IIα and topo IIβ. These exhibit a high degree of homology, except for some divergence in the C-terminal region. Both contain multiple bipartite nuclear localization sequences (NLS) that mediate their subnuclear localization. DNA Topoisomerase II Binding Protein 1 (TopBP1) binds the C-terminal region of Topo IIβ via its N-terminal region. In addition, TopBP1 contains a putative ADP-ribosylation site, two N-terminal NLS domains, and 8 repeating BRCA1 C-terminal (BRCT) domains found in DNA repair proteins, such as BRCA1, XRCC1, and Rad4. The BRCT domains of TopBP1 have been shown to bind DNA breaks, DNA nicks, and DNA termini, but not circular intact DNA. Thus, TopBP1 may localize topoisomerases to sites of DNA damage.
Host Species: Mouse
Clone: 33
Isotype: IgG1
Species Reactivity [for Features Main]: Human
Immunogen: Human TopBP1 aa. 204-416
Immunofluorescence, Western Blotting

Specifications
Specifications
| Antigen | TopBP1 |
| Applications | Western Blot |
| Classification | Monoclonal |
| Clone | 33 |
| Concentration | 250μg/mL |
| Conjugate | Unconjugated |
| Description | Topoisomerase II Binding Protein 1 |
| Formulation | Aqueous buffered solution containing BSA, glycerol, and ≤0.09% sodium azide. |
| Host Species | Mouse |
| Immunogen | Human TopBP1 aa. 204-416 |
| Show More |
For Research Use Only.
By clicking Submit, you acknowledge that you may be contacted by Fisher Scientific in regards to the feedback you have provided in this form. We will not share your information for any other purposes. All contact information provided shall also be maintained in accordance with our Privacy Policy.