Learn More
Description
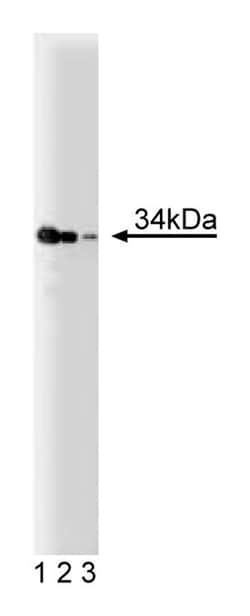
TNFα (Tumor Necrosis Factor) stimulates programmed cell death and NF-κB activation as a result of its binding to the TNF receptor 1 (TNFR1). Within this receptor, a sequence referred to as the “death domain” has been shown to be necessary for both of these functions. Using the yeast two-hybrid system to detect proteins which interact with the receptor through this “death domain”, a 34kDa protein was found and designated TRADD (TNFR1-Associated Death Domain protein). TRADD appears to contain no intrinsic catalytic activity. It also contains a death domain and it has been shown to bind to FADD and RIP. Mutational analysis of TRADD demonstrates that programmed cell death and NF-κB activation are distinct and controlled independently.
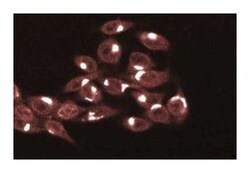
Immunofluorescence, Immunohistochemistry, Immunoprecipitation, Western Blotting

Specifications
Specifications
| Antigen | TRADD |
| Applications | Western Blot |
| Classification | Monoclonal |
| Clone | 37 |
| Concentration | 250μg/mL |
| Conjugate | Unconjugated |
| Description | TNFR1-Associated Death Domain protein |
| Formulation | Aqueous buffered solution containing BSA, glycerol, and ≤0.09% sodium azide. |
| Host Species | Mouse |
| Immunogen | Aqueous buffered solution containing BSA, glycerol, and ≤0.09% sodium azide. |
| Show More |
For Research Use Only.
By clicking Submit, you acknowledge that you may be contacted by Fisher Scientific in regards to the feedback you have provided in this form. We will not share your information for any other purposes. All contact information provided shall also be maintained in accordance with our Privacy Policy.