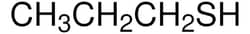Hydrocarbon derivatives
- (2)
- (2)
- (2)
- (2)
- (1)
- (3)
- (2)
- (1)
- (1)
- (2)
- (2)
- (2)
- (2)
- (2)
Filtered Search Results
1,4-Dithioerythritol, Electrophoresis Grade, 99%
CAS: 6892-68-8 Molecular Formula: C4H10O2S2 Molecular Weight (g/mol): 154.24 MDL Number: MFCD00063750 InChI Key: VHJLVAABSRFDPM-ZXZARUISSA-N Synonym: dithioerythritol,1,4-dithioerythritol,dte,2r,3s-1,4-dimercaptobutane-2,3-diol,erythro-1,4-dimercapto-2,3-butanediol,2,3-butanediol, 1,4-dimercapto-, 2r,3s-rel,1,4-dithioerythritol dte,2r*,3s*-1,4-dimercapto-2,3-butanediol,erythro-2,3-dihydroxy-1,4-butanedithiol,2r,3s-1,4-disulfanylbutane-2,3-diol PubChem CID: 439352 ChEBI: CHEBI:17456 SMILES: O[C@@H](CS)[C@H](O)CS
| PubChem CID | 439352 |
|---|---|
| CAS | 6892-68-8 |
| Molecular Weight (g/mol) | 154.24 |
| ChEBI | CHEBI:17456 |
| MDL Number | MFCD00063750 |
| SMILES | O[C@@H](CS)[C@H](O)CS |
| Synonym | dithioerythritol,1,4-dithioerythritol,dte,2r,3s-1,4-dimercaptobutane-2,3-diol,erythro-1,4-dimercapto-2,3-butanediol,2,3-butanediol, 1,4-dimercapto-, 2r,3s-rel,1,4-dithioerythritol dte,2r*,3s*-1,4-dimercapto-2,3-butanediol,erythro-2,3-dihydroxy-1,4-butanedithiol,2r,3s-1,4-disulfanylbutane-2,3-diol |
| InChI Key | VHJLVAABSRFDPM-ZXZARUISSA-N |
| Molecular Formula | C4H10O2S2 |
Thermo Scientific Chemicals DL-1,4-Dithiothreitol, 99%, for biochemistry
CAS: 12-3-3483 MDL Number: MFCD00004877 InChI Key: VHJLVAABSRFDPM-IMJSIDKUSA-N Synonym: dithiothreitol,dl-1,4-dithiothreitol,dl-dithiothreitol,1,4-dithio-dl-threitol,d-1,4-dithiothreitol,d-dtt,2s,3s-1,4-dimercaptobutane-2,3-diol,threo-1,4-dimercapto-2,3-butanediol,1,4-dithiothreitol,dtt PubChem CID: 446094 ChEBI: CHEBI:42170 IUPAC Name: (2S,3S)-1,4-bis(sulfanyl)butane-2,3-diol SMILES: C(C(C(CS)O)O)S
| PubChem CID | 446094 |
|---|---|
| CAS | 12-3-3483 |
| ChEBI | CHEBI:42170 |
| MDL Number | MFCD00004877 |
| SMILES | C(C(C(CS)O)O)S |
| Synonym | dithiothreitol,dl-1,4-dithiothreitol,dl-dithiothreitol,1,4-dithio-dl-threitol,d-1,4-dithiothreitol,d-dtt,2s,3s-1,4-dimercaptobutane-2,3-diol,threo-1,4-dimercapto-2,3-butanediol,1,4-dithiothreitol,dtt |
| IUPAC Name | (2S,3S)-1,4-bis(sulfanyl)butane-2,3-diol |
| InChI Key | VHJLVAABSRFDPM-IMJSIDKUSA-N |
1-Propanethiol 98.0+%, TCI America™
CAS: 107-03-9 Molecular Formula: C3H8S Molecular Weight (g/mol): 76.157 MDL Number: MFCD00004900 InChI Key: SUVIGLJNEAMWEG-UHFFFAOYSA-N Synonym: 1-propanethiol,propanethiol,n-propylmercaptan,propyl mercaptan,n-propyl mercaptan,n-propylthiol,1-propylmercaptan,propylthiol,1-mercaptopropane,1-propyl mercaptan PubChem CID: 7848 ChEBI: CHEBI:8473 IUPAC Name: propane-1-thiol SMILES: CCCS
| PubChem CID | 7848 |
|---|---|
| CAS | 107-03-9 |
| Molecular Weight (g/mol) | 76.157 |
| ChEBI | CHEBI:8473 |
| MDL Number | MFCD00004900 |
| SMILES | CCCS |
| Synonym | 1-propanethiol,propanethiol,n-propylmercaptan,propyl mercaptan,n-propyl mercaptan,n-propylthiol,1-propylmercaptan,propylthiol,1-mercaptopropane,1-propyl mercaptan |
| IUPAC Name | propane-1-thiol |
| InChI Key | SUVIGLJNEAMWEG-UHFFFAOYSA-N |
| Molecular Formula | C3H8S |
eMolecules Chem-Impex Fmoc-N-methyl-S-tert-butylthio-L-cysteine 250mg 272389673 29640 0 000 1013096-03-1 445 590 C23H27NO4S2
Chem-Impex Fmoc-N-methyl-S-tert-butylthio-L-cysteine 250mg 272389673 29640 0 000 1013096-03-1 445 590 C23H27NO4S2

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Sigma Aldrich 1-Propanethiol
MilliporeSigma Sigma Organics products encompass a wide range of quality reagents, solvents, catalysts, and building blocks for organic synthesis. From benchtop discovery to process development and scale-up, Sigma Organics solutions are built to meet the needs of synthetic chemists.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
| Boiling Point | 67°C to 68°C |
|---|---|
| Linear Formula | CH3CH2CH2SH |
| Molecular Weight (g/mol) | 76.16 |
| Density | 0.820 g/mL (at 25°C) |
| Percent Purity | 99% |
| CAS | 107-03-9 |
| MDL Number | MFCD00004900 |
| Refractive Index | n20/D 1.426 (literature) |
| Synonym | 1-PP; n-Propylmercaptan; Mercaptan C3 |
| RTECS Number | TZ7300000 |
| Recommended Storage | Room Temperature |
| Molecular Formula | C3H8S |
| EINECS Number | 203-455-5 |
| Melting Point | -113°C |
Medchemexpress LLC 9-Decyn-1-ol | 17643-36-6 | 100 G
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
9-Decyn-1-ol is an alkyl/ether-based PROTAC linker used in the synthesis of PROTACs. It can be employed to conjugate GDC-0068 with Lenalidomide, resulting in INY-03-041, a potent, highly selective, and PROTAC-based pan-Akt degrader. It also functions as a click chemistry reagent.
- Used in the synthesis of PROTACs
- Can conjugate GDC-0068 with Lenalidomide to create INY-03-041, a pan-Akt degrader
- Inhibits Akt1, Akt2, and Akt3 with IC50s of 2.0 nM, 6.8 nM, and 3.5 nM, respectively
- Click chemistry reagent containing an Alkyne group
- Undergoes copper-catalyzed azide-alkyne cycloaddition (CuAAc) with molecules containing Azide groups

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC 9-Decyn-1-ol | 17643-36-6 | 50 G
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
9-Decyn-1-ol is an alkyl/ether-based PROTAC linker used in the synthesis of PROTACs. It can conjugate GDC-0068 with Lenalidomide to create INY-03-041, a potent, highly selective, and PROTAC-based pan-Akt degrader. INY-03-041 inhibits Akt1, Akt2, and Akt3 with IC50s of 2.0 nM, 6.8 nM, and 3.5 nM, respectively. It is also a click chemistry reagent, containing an Alkyne group that can undergo copper-catalyzed azide-alkyne cycloaddition (CuAAc) with molecules containing Azide groups.
- Alkyl/ether-based PROTAC linker
- Used in the synthesis of PROTACs
- Generates a potent, highly selective, PROTAC-based pan-Akt degrader
- Click chemistry reagent with an alkyne group
- Undergoes copper-catalyzed azide-alkyne cycloaddition (CuAAc) with azide groups

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More