Learn More
FAK (pY397) (Focal Adhesion Kinase) Mouse anti-Human, Unlabeled, Clone: 14, BD
Mouse Monoclonal Antibody
$462.50 - $732.00
Specifications
| Antigen | FAK (pY397) (Focal Adhesion Kinase) |
|---|---|
| Clone | 14 |
| Concentration | 250μg/mL |
| Applications | Western Blot |
| Classification | Monoclonal |
Description
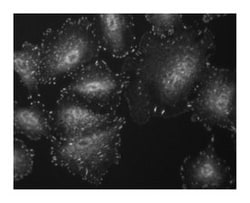
Focal Adhesion Kinase (FAK) is a cytoplasmic tyrosine kinase that colocalizes with integrins in focal adhesions. This cellular localization is directed by a 125 amino acid sequence at the C-terminus called the “Focal Adhesion Targeting” sequence (FAT). The binding of extracellular matrix ligands to integrins triggers autophosphorylation at Tyr-397, and activation of FAK through phosphorylation of Tyr residues (Tyr-576 and Tyr577) in the kinase domain activation loop. For example, cell adhesion to a fibronectin substratum involves concurrent activation of Src and phosphorylation of the FAK activation loop. In addition, phosphorylation of other Tyr residues (Tyr-925, and Tyr-861) creates binding sites for SH2 domains of intracellular signaling molecules such as Src, PI3 kinase, and Grb2. FAK's ability to bind numerous structural and signaling proteins via a variety of interactions is important for FAK activation level, and for FAK interaction with a variety of substrates localized to sites of cell adhesion. Thus, FAK activity is regulated by a complex set of phosphorylation sites, and thisphospho-regulation could be important for cell motility, cell growth, cytoskeletal organization, and adhesion-dependent cell survival.
Host Species: Mouse
Clone: 14
Isotype: IgG1
Species Reactivity [for Features Main]: Human
Immunogen: Human FAK (pY397) Peptide
Immunofluorescence, Western Blotting
Specifications
| FAK (pY397) (Focal Adhesion Kinase) | |
| 250μg/mL | |
| Monoclonal | |
| Mouse | |
| Cell Biology | |
| Aqueous buffered solution containing BSA, glycerol, and ≤0.09% sodium azide. | |
| IgG1 | |
| Affinity Purified | |
| Focal Adhesion Kinase (pY397) |
| 14 | |
| Western Blot | |
| Unconjugated | |
| RUO | |
| Human | |
| Human FAK (pY397) Peptide | |
| Primary | |
| Store undiluted at -20°C. |
For Research Use Only.
Your input is important to us. Please complete this form to provide feedback related to the content on this product.