Learn More
PAI-1 Mouse, Unlabeled, Clone: 41, BD
Mouse Monoclonal Antibody
$286.00 - $566.00
Specifications
| Antigen | PAI-1 |
|---|---|
| Clone | 41 |
| Concentration | 250μg/mL |
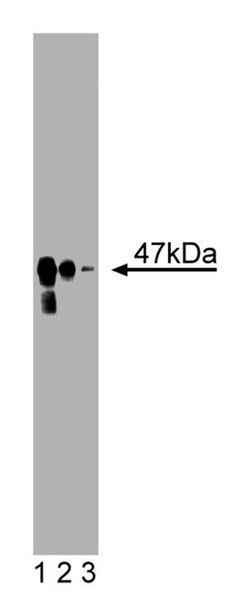
| Applications | Western Blot |
| Classification | Monoclonal |
Description
Fibrinolysis is regulated by the plasminogen activators, tissue plasminogen activator (tPA) and urokinase PA (uPA), and by the plasminogen activator-inhibitors (PAIs). Two PAIs include the serpin family members, PAI-1 and PAI-2. PAI-1 is a glycoprotein found in plasma, platelets, endothelial cells, hepatoma cells, and fibrosarcoma cells. Thrombin, endotoxin, and IL-1 induce PAI-1 synthesis in endothelial cells, which is where the major portion of plasma PAI-1 is produced. PAI-2 is a glycoprotein expressed in placenta and monocyte macrophages. The uPA/plasmin system may play a key role in cancer progression through degradation of the extracellular matrix during tumor cell migration. Paradoxically, high levels of PAI-1 are also predictive of poor prognosis of cancer patients. This finding may be a result of the role of plasmin proteolysis in the prevention of tumor vessel assembly. PAI-1 can promote tumor angiogenesis, and the mechanism may involve PAI-induced regulation of plasmin proteolysis during tumor angiogenesis. Thus, PAI-1 is a serpin protease inhibitor that is important for the regulation of plasmin proteolysis during fibrinolysis and extracellular matrix degradation.
Host Species: Mouse
Clone: 41
Isotype: IgG1
Species Reactivity [for Features Main]: Human
Immunogen: Human PAI-1 aa. 207-329
Immunofluorescence, Western Blotting
Specifications
| PAI-1 | |
| 250μg/mL | |
| Monoclonal | |
| Mouse | |
| Cell Biology | |
| Aqueous buffered solution containing BSA, glycerol, and ≤0.09% sodium azide. | |
| IgG1 | |
| Affinity Purified |
| 41 | |
| Western Blot | |
| Unconjugated | |
| RUO | |
| Canine, Chicken, Human, Mouse, Rat | |
| Human PAI-1 aa. 207-329 | |
| Primary | |
| Store undiluted at -20°C. |
For Research Use Only.
Your input is important to us. Please complete this form to provide feedback related to the content on this product.