Promotional price valid on web orders only. Your contract pricing may differ. Interested in signing up for a dedicated account number?
Learn More
Learn More
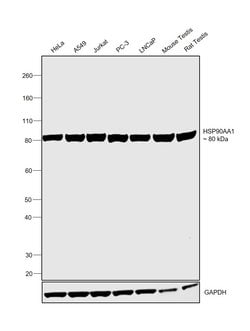
Invitrogen™ HSP90 alpha Monoclonal Antibody (OTI3B5)
Mouse Monoclonal Antibody
Supplier: Invitrogen™ MA525036
Description
HSP90 alpha Monoclonal Antibody for Western Blot, ICC/IF
Heat shock proteins (HSP) are proteins that are expressed in response to various biological stress, including heat. HSP90 is a 90 kDa protein that is induced under stress conditions, but is also one of the most abundant cellular proteins found under non-stress conditions. HSP90 has been found to be associated with a number of other intracellular proteins, including steroid receptors, actin, tubulin, and some kinases. It has been suggested that HSP90 is necessary to maintain structural integrity of at least some steroid receptors to allow for ligand binding during receptor activation.
Specifications
| HSP90 alpha | |
| Monoclonal | |
| 1 mg/mL | |
| PBS with 1% BSA, 50% glycerol and 0.02% sodium azide; pH 7.3 | |
| P07900 | |
| Hsp90aa1 | |
| Full length human recombinant protein of HSP90AA1 produced in HEK293T cell. | |
| 100 μL | |
| Primary | |
| Human | |
| Antibody | |
| IgG1 |
| Western Blot, Immunocytochemistry | |
| OTI3B5 | |
| Unconjugated | |
| Hsp90aa1 | |
| 86kDa; 89kDa; 90-kDa heat shock protein; 90-kDa heat shock protein alpha; akineto; AL024080; AL024147; EL52; epididymis luminal secretory protein 52; epididymis secretory sperm binding protein Li 65p; fb17b01; heat shock 86 kDa; heat shock 90kD protein 1, alpha; heat shock 90kD protein 1, alpha-like 4; heat shock 90kD protein, alpha-like 4; heat shock 90kDa protein 1, alpha; heat shock 90kDa protein 1, beta; Heat shock protein; heat shock protein 1, alpha; heat shock protein 86; heat shock protein 90; heat shock protein 90 alpha family class A member 1; heat shock protein 90, alpha (cytosolic), class A member 1; heat shock protein 90, alpha (cytosolic), class A member 1, tandem duplicate 1; heat shock protein 90-alpha 1; heat shock protein 90kDa alpha (cytosolic), class A member 1; heat shock protein 90kDa alpha family class A member 1; heat shock protein alpha; Heat shock protein HSP 90-alpha; Heat shock protein HSP 90-alpha 1; heat shock protein HSP90 alpha; heat shock protein hsp90alpha; heat shock protein, 1; heat shock protein, 86 kDa 1; heat shock protein, 89 kDa; Heat shock protein90; HEL-S-65p; HSP; HSP 86; HSP 90 alpha; hsp4; HSP86; Hsp86-1; Hsp89; HSP89A; HSP90; hsp90a; hsp90a protein; hsp90a.1; hsp90aa1; hsp90aa1.1; hsp90alpha; HSP90N; HSPC1; Hspca; HSPCA protein; HSPCAL1; HSPCAL4; HSPN; hypothetical protein; LAP2; LAP-2; Lipopolysaccharide-associated protein 2; LPS-associated protein 2; renal carcinoma antigen NY-REN-38; slo; sloth; TSTA; Tumor-specific transplantation 86 kDa antigen; wu:fb17b01; zgc:86652 | |
| Mouse | |
| Affinity chromatography | |
| RUO | |
| 3320 | |
| -20°C, Avoid Freeze/Thaw Cycles | |
| Liquid |
Product Content Correction
Your input is important to us. Please complete this form to provide feedback related to the content on this product.
Product Title
Spot an opportunity for improvement?Share a Content Correction