Life Science Buffers
Balanced salt solutions and powders used for molecular, protein, nucleic acid, and cell biology applications including cell culture, electrophoresis, ELISA, and chromatography processes; available in a variety of formulations and grades.
Buffer solutions serve to keep pH (acidity or alkalinity) nearly constant in a variety of chemical and biological applications. For example, a bicarbonate buffer helps maintain the pH of blood, a buffered saline maintains cellular contents at a consistent pH level, and buffers help maintain a narrow pH range for enzymes to function correctly.
Most biological samples used in research are kept at a pH of about 7.4. Common biological buffers used for tissue culture include Dulbeccos phosphate buffered saline (PBS), Tris base, HEPES, MOPS, PIPES, and other formulations developed for specific cell lines or applications. Most buffers used in cell culture are also DNase-, RNase-, and protease-free.
The choice of buffer for a particular biological reaction or site depends on several factors:
- Temperature
- Desired or target pH
- Buffer toxicity (to the system)
- Buffer interactions with other system components
- (1)
- (1)
- (3)
- (1)
- (108)
- (1)
- (2)
- (1)
- (2)
- (2)
- (69)
- (2)
- (1)
- (1)
- (1)
- (1)
- (10)
- (3)
- (2)
- (10)
- (6)
- (1)
- (4)
- (2)
- (10)
- (1)
- (5)
- (5)
- (110)
- (107)
- (1)
- (1)
- (1)
- (1)
- (1)
- (11)
- (2)
- (7)
- (1)
- (1)
- (1)
- (2)
- (6)
- (8)
- (23)
- (1)
- (12)
- (22)
- (1)
- (4)
- (1)
- (12)
- (1)
- (3)
- (1)
- (53)
- (6)
- (5)
- (34)
- (129)
- (2)
- (1)
- (26)
- (2)
- (1)
- (6)
- (1)
- (1)
- (3)
- (19)
- (2)
- (1)
- (26)
- (1)
- (1)
- (6)
- (7)
- (3)
- (8)
- (1)
- (86)
- (148)
- (8)
- (6)
- (10)
- (1)
- (1)
- (4)
- (1)
- (1)
- (1)
- (1)
- (1)
- (2)
- (2)
- (17)
- (143)
- (6)
- (3)
- (1)
- (3)
- (3)
- (7)
- (5)
- (3)
- (8)
- (1)
- (598)
- (5)
- (4)
- (4)
- (114)
- (23)
- (12)
- (48)
- (5)
- (4)
- (2)
- (5)
- (125)
- (2)
- (13)
- (10)
- (3)
- (14)
- (49)
- (1)
- (2)
- (31)
- (13)
- (26)
- (2)
- (7)
- (9)
- (9)
- (23)
- (45)
- (5)
- (41)
- (11)
- (5)
- (13)
- (1)
- (3)
- (2)
- (3)
- (1)
- (9)
- (1)
- (94)
- (3)
- (4)
- (4)
- (3)
- (3)
- (3)
- (10)
- (3)
- (31)
- (4)
- (3)
- (9)
- (3)
- (45)
- (1)
- (3)
- (1)
- (4)
- (1)
- (1)
- (2)
- (1)
- (1)
- (3)
- (5)
- (1)
- (4)
- (3)
- (5)
- (1)
- (10)
- (10)
- (6)
- (4)
- (2)
- (5)
- (3)
- (2)
- (5)
- (10)
- (1)
- (10)
- (1)
- (2)
- (3)
- (2)
- (2)
- (2)
- (5)
- (7)
- (1)
- (2)
- (10)
- (4)
- (2)
- (1)
- (1)
- (4)
- (3)
- (3)
- (1)
- (2)
- (1)
- (3)
- (3)
- (2)
- (13)
- (4)
- (1)
- (4)
- (3)
- (2)
- (2)
- (2)
- (1)
- (4)
- (2)
- (4)
- (3)
- (10)
- (1)
- (1)
- (1)
- (1)
- (14)
- (1)
- (38)
- (2)
- (1)
- (2)
- (9)
- (2)
- (3)
- (8)
- (2)
- (1)
- (2)
- (3)
- (1)
- (1)
- (1)
- (32)
- (1)
- (5)
- (8)
- (2)
- (14)
- (5)
- (1)
- (3)
- (5)
- (1)
- (23)
- (1)
- (62)
- (11)
- (1)
- (43)
- (1)
- (11)
- (2)
- (1)
- (2)
- (1)
- (9)
- (1)
- (38)
- (1)
- (1)
- (7)
- (1)
- (1)
- (5)
- (1)
- (2)
- (1)
- (1)
- (21)
- (5)
- (3)
- (1)
- (17)
- (3)
- (1)
- (4)
- (4)
- (1)
- (6)
- (1)
- (4)
- (77)
- (93)
- (3)
- (6)
- (21)
- (53)
- (1)
- (72)
- (1)
- (29)
- (1)
- (1)
- (49)
- (15)
- (5)
- (2)
- (29)
- (1)
- (1)
- (10)
- (5)
- (5)
- (4)
- (2)
- (1)
- (7)
- (12)
- (2)
- (7)
Filtered Search Results
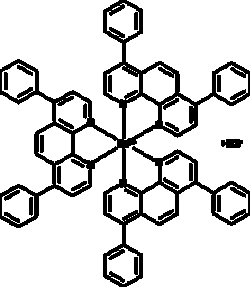
Cayman Chemical TrIs 4 7dIphenyl1 10phen 25mg
A luminescent probe (absorption λmax: 455 nm, luminescence λmax: 613 nm) widely used for detection and quantitation of oxygen; strongly reduced by molecular oxygen as a result of dynamic quenching; thus oxygen detection can be based on either measurement of intensity or decay time

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Teknova 1X PBS with 4mM EDTA, pH 7.4. 1000mL, Sterile.
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Phosphate-buffered saline (PBS) is a salt solution that is commonly used in physiological and biochemical experiments. It can be used to maintain pH neutrality or to buffer against changes in pH in order to provide optimal conditions for cellular growth and function. The buffering range is between pH 6.8 and 7.4.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Teknova 1M Tris-HCl, pH 8.0. 500mL, Sterile.
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Tris (tris(hydroxymethyl)aminomethane) is used as a component ofbuffer solutions, especially for solutions ofnucleic acids. The useful buffer range for tris (pH 7–9) is similar to the pH of most living organisms. This, and its low cost, make tris one of the most common buffers in the biology/biochemistry laboratory.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Teknova TE Buffer (10mM Tris, 1mM EDTA), pH 8.0. 1mL, 50-Pack, Sterile. DNase/RNase/PCR
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
TE buffer is a salt solution that is commonly used to resuspend (or dilute) DNA and RNA. Composed of Tris base and EDTA, these molecules work together to maintain pH and prevent nucleic acids from degradation.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Teknova 10X PBS with 0.5% Tween 20. 1000mL, Sterile.
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Phosphate-buffered saline (PBS) is a salt solution that is commonly used in physiological and biochemical experiments. It can be used to maintain pH neutrality or to buffer against changes in pH in order to provide optimal conditions for cellular growth and function. The buffering range is between pH 6.8 and 7.4.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Teknova PPB Solution. 1000mL, Sterile.
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
30mM Tris-HCl pH 8.0, 1mM Ethylenediaminetetraacetic acid (EDTA) pH 8.0,20% Sucrose

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Teknova 1M Tris-HCl, pH 7.2 Solution. 1000mL, Sterile.
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Tris (tris(hydroxymethyl)aminomethane) is used as a component ofbuffer solutions, especially for solutions ofnucleic acids. The useful buffer range for tris (pH 7–9) is similar to the pH of most living organisms. This, and its low cost, make tris one of the most common buffers in the biology/biochemistry laboratory.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Teknova 1M Tris-HCl, pH 8.5. 1000 mL, Sterile.
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Tris (tris(hydroxymethyl)aminomethane) is used as a component ofbuffer solutions, especially for solutions ofnucleic acids. The useful buffer range for tris (pH 7–9) is similar to the pH of most living organisms. This, and its low cost, make tris one of the most common buffers in the biology/biochemistry laboratory.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Teknova 1M Tris-HCl, pH 9.0. 1000mL, Sterile.
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Tris (tris(hydroxymethyl)aminomethane) is used as a component ofbuffer solutions, especially for solutions ofnucleic acids. The useful buffer range for tris (pH 7–9) is similar to the pH of most living organisms. This, and its low cost, make tris one of the most common buffers in the biology/biochemistry laboratory.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Teknova DNA Suspension Buffer, pH 8.0. RNase/DNase Tested, PCR Grade, 1L, Sterile. (10mM Tris, 0.1mM EDTA)
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
10mM Tris-HCl, 0.1mM Ethylenediaminetetraacetic acid (EDTA)

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Teknova 1M Tris-HCl, pH 7.0, 500ml, Sterile.
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Tris (tris(hydroxymethyl)aminomethane) is used as a component ofbuffer solutions, especially for solutions ofnucleic acids. The useful buffer range for tris (pH 7–9) is similar to the pH of most living organisms. This, and its low cost, make tris one of the most common buffers in the biology/biochemistry laboratory.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Teknova DNA Suspension Buffer, pH 8.0. 100mL, Sterile. DNase/RNase Tested, PCR Grade.
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
TE buffer is a salt solution that is commonly used to resuspend (or dilute) DNA and RNA. Composed of Tris base and EDTA, these molecules work together to maintain pH and prevent nucleic acids from degradation.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Teknova 1X PBS with 0.05% Tween 20, pH 7.4. 1000mL, Sterile.
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Phosphate-buffered saline (PBS) is a salt solution that is commonly used in physiological and biochemical experiments. It can be used to maintain pH neutrality or to buffer against changes in pH in order to provide optimal conditions for cellular growth and function. The buffering range is between pH 6.8 and 7.4.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Teknova 1M Tris-HCl, pH 8.0 Solution. 1000mL, Sterile.
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Tris (tris(hydroxymethyl)aminomethane) is used as a component ofbuffer solutions, especially for solutions ofnucleic acids. The useful buffer range for tris (pH 7–9) is similar to the pH of most living organisms. This, and its low cost, make tris one of the most common buffers in the biology/biochemistry laboratory.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Teknova 1M Tris-HCl, pH 8.3, 1000mL, Sterile.
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Tris (tris(hydroxymethyl)aminomethane) is used as a component ofbuffer solutions, especially for solutions ofnucleic acids. The useful buffer range for tris (pH 7–9) is similar to the pH of most living organisms. This, and its low cost, make tris one of the most common buffers in the biology/biochemistry laboratory.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More