Promotional price valid on web orders only. Your contract pricing may differ. Interested in signing up for a dedicated account number?
Learn More
Learn More
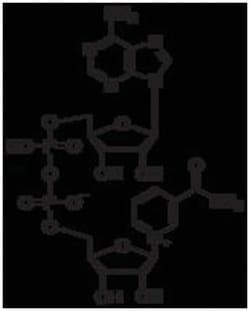
MP Biomedicals™ β-Nicotinamide Adenine Dinucleotide, Oxidized Form, MP Biomedicals™
Click to view available options
Quantity:
100 mg
500 mg
Description
β-NAD, a pyridine nucleotide and biologically active form of nicotinic acid, is a coenzyme necessary for the catalytic reaction of certain enzymes. It occurs in living cells primarily in the oxidized state. It serves as a coenzyme of the dehydrogenases, especially in the dehydrogenation of primary and secondary alcohols. NAD usually acts as a hydrogen acceptor, forming NADH which then serves as a hydrogen donor in the respiratory chain.
- Electron acceptor.
- β-NAD is a carrier for the hydride ion, forming b-NADH.
- Hydride ion is enzymatically removed from a substrate molecule by the action of dehydrogenases such as malic dehydrogenase and lactic dehydrogenase.
- Such enzymes catalyze the reversible transfer of a hydride ion from malate or lactate to b-NAD to form the reduced product, b-NADH.
- Unlike b-NAD which has no absorbance at 340nm, b-NADH absorbs at 340nm (EmM = 6.22).
- The increase in absorbance at 340nm with the formation of b-NADH is the basis for measurement of activity of many enzymes.

Specifications
Specifications
| Grade | Cell Culture |
| Purity | ≥98% |
| pH | 2 to 4 (1% aq soln) |
| Content And Storage | 0°C, Desiccate |
| Quantity | 100 mg |
| Molecular Weight (g/mol) | 663.4 (anhyd.) |
Safety and Handling
WARNING: Reproductive Harm - www.P65Warnings.ca.gov
Product Content Correction
Your input is important to us. Please complete this form to provide feedback related to the content on this product.
Product Title
Spot an opportunity for improvement?Share a Content Correction