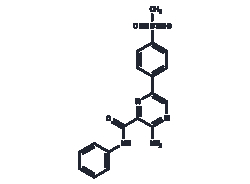
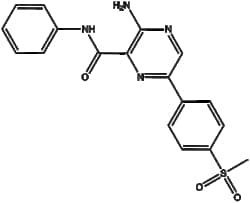
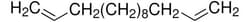Unsaturated hydrocarbons
- (1)
- (4)
- (1)
- (1)
- (1)
- (1)
- (1)
- (4)
- (3)
- (1)
- (1)
- (1)
- (1)
- (5)
- (1)
- (1)
- (2)
Filtered Search Results
1,2,3,4,5-Pentamethylcyclopentadiene, 94%
CAS: 4045-44-7 Molecular Formula: C10H16 Molecular Weight (g/mol): 136.238 MDL Number: MFCD00001354 InChI Key: WQIQNKQYEUMPBM-UHFFFAOYSA-N Synonym: 1,2,3,4,5-pentamethylcyclopentadiene,pentamethylcyclopentadiene,cyclopentadiene, 1,2,3,4,5-pentamethyl,1,3-cyclopentadiene, 1,2,3,4,5-pentamethyl,1,3-cyclopentadiene, pentamethyl,1,2,3,4,5-pentamethyl-1,3-cyclopentadiene,pubchem20101,acmc-209je4,wqiqnkqyeumpbm-uhfffaoysa,1,2,3,4,5-pentamethylcyclopentadien PubChem CID: 77667 IUPAC Name: 1,2,3,4,5-pentamethylcyclopenta-1,3-diene SMILES: CC1C(=C(C(=C1C)C)C)C
| PubChem CID | 77667 |
|---|---|
| CAS | 4045-44-7 |
| Molecular Weight (g/mol) | 136.238 |
| MDL Number | MFCD00001354 |
| SMILES | CC1C(=C(C(=C1C)C)C)C |
| Synonym | 1,2,3,4,5-pentamethylcyclopentadiene,pentamethylcyclopentadiene,cyclopentadiene, 1,2,3,4,5-pentamethyl,1,3-cyclopentadiene, 1,2,3,4,5-pentamethyl,1,3-cyclopentadiene, pentamethyl,1,2,3,4,5-pentamethyl-1,3-cyclopentadiene,pubchem20101,acmc-209je4,wqiqnkqyeumpbm-uhfffaoysa,1,2,3,4,5-pentamethylcyclopentadien |
| IUPAC Name | 1,2,3,4,5-pentamethylcyclopenta-1,3-diene |
| InChI Key | WQIQNKQYEUMPBM-UHFFFAOYSA-N |
| Molecular Formula | C10H16 |
Tetramethylcyclopentadiene, mixed isomers, tech. 85%
CAS: 4249-10-9 Molecular Formula: C9H14 Molecular Weight (g/mol): 122.21 MDL Number: MFCD00145385 InChI Key: VNPQQEYMXYCAEZ-UHFFFAOYSA-N Synonym: 1,2,3,4-tetramethylcyclopentadiene,tetramethylcyclopentadiene,1,2,3,4-tetramethyl-1,3-cyclopentadiene,tetramethyl cyclopentadiene,tetramethylcyclopentadiene, mixed isomers,pubchem15072,acmc-1an6q,1,3-cyclopentadiene, tetramethyl,2,3,4,5-tetramethylcyclopenta-2,4-diene PubChem CID: 138163 IUPAC Name: 1,2,3,4-tetramethylcyclopenta-1,3-diene SMILES: CC1=C(C(=C(C1)C)C)C
| PubChem CID | 138163 |
|---|---|
| CAS | 4249-10-9 |
| Molecular Weight (g/mol) | 122.21 |
| MDL Number | MFCD00145385 |
| SMILES | CC1=C(C(=C(C1)C)C)C |
| Synonym | 1,2,3,4-tetramethylcyclopentadiene,tetramethylcyclopentadiene,1,2,3,4-tetramethyl-1,3-cyclopentadiene,tetramethyl cyclopentadiene,tetramethylcyclopentadiene, mixed isomers,pubchem15072,acmc-1an6q,1,3-cyclopentadiene, tetramethyl,2,3,4,5-tetramethylcyclopenta-2,4-diene |
| IUPAC Name | 1,2,3,4-tetramethylcyclopenta-1,3-diene |
| InChI Key | VNPQQEYMXYCAEZ-UHFFFAOYSA-N |
| Molecular Formula | C9H14 |
1,13-Tetradecadiene, 90+%
CAS: 21964-49-8 Molecular Formula: C14H26 Molecular Weight (g/mol): 194.362 MDL Number: MFCD00014941 InChI Key: XMRSTLBCBDIKFI-UHFFFAOYSA-N Synonym: 1,13-tetradecadiene,tetradecadiene-1,13,acmc-209fpz PubChem CID: 30875 IUPAC Name: tetradeca-1,13-diene SMILES: C=CCCCCCCCCCCC=C
| PubChem CID | 30875 |
|---|---|
| CAS | 21964-49-8 |
| Molecular Weight (g/mol) | 194.362 |
| MDL Number | MFCD00014941 |
| SMILES | C=CCCCCCCCCCCC=C |
| Synonym | 1,13-tetradecadiene,tetradecadiene-1,13,acmc-209fpz |
| IUPAC Name | tetradeca-1,13-diene |
| InChI Key | XMRSTLBCBDIKFI-UHFFFAOYSA-N |
| Molecular Formula | C14H26 |
1,13-Tetradecadiene 90.0+%, TCI America™
CAS: 21964-49-8 Molecular Formula: C14H26 Molecular Weight (g/mol): 194.362 MDL Number: MFCD00014941 InChI Key: XMRSTLBCBDIKFI-UHFFFAOYSA-N Synonym: 1,13-tetradecadiene,tetradecadiene-1,13,acmc-209fpz PubChem CID: 30875 IUPAC Name: tetradeca-1,13-diene SMILES: C=CCCCCCCCCCCC=C
| PubChem CID | 30875 |
|---|---|
| CAS | 21964-49-8 |
| Molecular Weight (g/mol) | 194.362 |
| MDL Number | MFCD00014941 |
| SMILES | C=CCCCCCCCCCCC=C |
| Synonym | 1,13-tetradecadiene,tetradecadiene-1,13,acmc-209fpz |
| IUPAC Name | tetradeca-1,13-diene |
| InChI Key | XMRSTLBCBDIKFI-UHFFFAOYSA-N |
| Molecular Formula | C14H26 |
Medchemexpress LLC 8-bromooct-1-yne | 81216-13-9 | MFCD20621358 | 189.09 g·mol⁻¹ | C8H13Br | 1 G
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
8-Bromooct-1-yne is a brominated terminal alkyne reagent used as a synthetic building block in organic synthesis and chemical biology research. It combines a terminal alkyne for click-type reactions with a bromide leaving group for nucleophilic substitution, enabling installation of alkynyl linkers and chain elaboration in probe and linker synthesis.
- Terminal alkyne enables copper-catalyzed azide-alkyne cycloaddition (click chemistry).
- Bromide functions as a good leaving group for nucleophilic substitution and chain modification.
- Suitable as a building block for probes, tags, and linker or PROTAC synthesis.
- Available in small laboratory quantities for research use.
- Handle under inert atmosphere and store cold according to handling guidelines.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC MI-503 | 1857417-13-0 | 99.87% | 564.63 | 1 ML
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
MI-503 | 1857417-13-0 | 99.87% | 564.63 | 1 ML

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC 8-bromooct-1-yne | 81216-13-9 | MFCD20621358 | 189.09 | C8H13Br | 500 MG
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
8-Bromooct-1-yne is an organobromide reagent containing a terminal alkyne and a bromide leaving group. It is used as a synthetic building block in organic chemistry and for bioconjugation workflows such as copper-catalyzed azide-alkyne cycloaddition (click chemistry). Store under nitrogen at 4°C; in solution, keep at -80°C (up to 6 months) or -20°C (up to 1 month). CAS: 81216-13-9; formula: C8H13Br; MW: 189.09.
- Terminal alkyne for conjugation and cross-coupling reactions.
- Bromide leaving group enables nucleophilic substitution.
- Suitable for copper-catalyzed click chemistry with azide partners.
- Supplied in small-scale quantities for research applications.
- Store under inert atmosphere to preserve stability.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC 8-bromooct-1-yne | 81216-13-9 | MFCD20621358 | 189.09 g/mol | C8H13Br | 250 MG
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
8-Bromooct-1-yne is a terminal alkyne bearing a bromide substituent used as a synthetic building block for organic synthesis and chemical biology. It participates in alkyne-based coupling and substitution reactions, enabling installation of functional groups or linkers in small-molecule synthesis and bioconjugation.
- Terminal alkyne with a reactive bromide group.
- Compatible with copper-catalyzed azide-alkyne cycloaddition (click chemistry) and cross-coupling reactions.
- Useful as an alkylating building block for functionalization and chain extension.
- Provided as a 250 mg research reagent; store under inert atmosphere at low temperature for stability.
- Molecular formula C8H13Br; molecular weight 189.09 g/mol.
- Purity not specified on the manufacturer product page.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC 8-bromooct-1-yne | 81216-13-9 | MFCD20621358 | 189.09 g/mol | C8H13Br | 100 MG
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
8-Bromooct-1-yne is a brominated terminal alkyne reagent used in organic synthesis and copper-catalyzed click chemistry. It offers both a reactive alkyne for azide coupling and a bromide leaving group for substitution, supplied in small research-scale quantities and requiring inert storage.
- Terminal alkyne reagent for organic synthesis and click chemistry.
- Contains a bromide leaving group for nucleophilic substitution.
- Molecular formula C8H13Br; molecular weight 189.09 g/mol.
- Suitable for copper-catalyzed azide-alkyne cycloaddition (CuAAC).
- Available in small research-scale pack sizes for laboratory use.
- Store under nitrogen at 4°C; in solvent: -80°C (6 months) or -20°C (1 month).

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Sigma Aldrich 1,13-Tetradecadiene
MilliporeSigma Sigma Organics products encompass a wide range of quality reagents, solvents, catalysts, and building blocks for organic synthesis. From benchtop discovery to process development and scale-up, Sigma Organics solutions are built to meet the needs of synthetic chemists.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
| CAS | 21964-49-8 |
|---|
TARGETMOL CHEMICALS INC VE-821 25MG
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Also available in 2 mg 5 mg 10 mg 50 mg 100 mg 1 mL 10 mM (in DMSO) and bulk. Please contact Fisher for quotes. VE-821 (ATR Inhibitor IV) is a selective ATP competitive inhibitor of ATR( Ki/IC50 13/26 nM in cell-free assays). purity: 99%

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC VE-821 | 1232410-49-9 | 99.7% | 368.41 | 100 MG
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
VE-821 is a potent ATP-competitive inhibitor of ATR with Ki/IC50 values of 13 nM/26 nM. It exhibits excellent selectivity for ATR, demonstrating minimal cross-reactivity with related PIKKs like ATM, DNA-PK, mTOR, and PI3Kγ. This compound enhances the sensitivity of pancreatic cancer cells (PSN-1, MiaPaCa-2, and primary PancM) to radiation and Gemcitabine under both normoxic and hypoxic conditions. ATR inhibition by VE-821 also leads to the inhibition of radiation-induced G2/M arrest in cancer cells.
- Potent ATP-competitive inhibitor of ATR (Ki/IC50 of 13 nM/26 nM)
- Exhibits excellent selectivity for ATR with minimal cross-reactivity against related PIKKs
- Enhances sensitivity of pancreatic cancer cells to radiation and Gemcitabine
- Inhibits radiation-induced G2/M arrest in cancer cells
- Inhibits phosphorylation of Chk1 (Ser 345) after specific treatments

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC VE-821 | 1232410-49-9 | 99.7% | 368.41 | 25 MG
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
VE-821 is a potent ATP-competitive inhibitor of ATR with a Ki/IC50 of 13 nM/26 nM. It demonstrates excellent selectivity for ATR with minimal cross-reactivity against related PIKKs such as ATM, DNA-PK, mTOR, and PI3Kγ. This inhibitor also significantly enhances the sensitivity of PSN-1, MiaPaCa-2, and primary PancM pancreatic cancer cells to radiation and Gemcitabine, under both normoxic and hypoxic conditions. ATR inhibition by VE-821 leads to the inhibition of radiation-induced G2/M arrest in cancer cells.
- Potent ATP-competitive inhibitor of ATR
- Ki/IC50 of 13 nM/26 nM for ATR
- Demonstrates excellent selectivity for ATR with minimal cross-reactivity against related PIKKs
- Enhances sensitivity of pancreatic cancer cells to radiation and Gemcitabine
- Inhibits radiation-induced G2/M arrest in cancer cells
- Inhibits phosphorylation of Chk1 (Ser 345)

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC VE-821 | 1232410-49-9 | 99.7% | 368.41 | 50 MG
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
VE-821 is a potent ATP-competitive inhibitor of ATR, with Ki/IC50 values of 13 nM/26 nM. It demonstrates excellent selectivity for ATR, showing minimal cross-reactivity against related PIKKs such as ATM, DNA-PK, mTOR, and PI3Kγ.
- Shows minimal cross-reactivity against related PIKKs ATM, DNA-PK, mTOR, and PI3Kγ
- Significantly enhances the sensitivity of PSN-1, MiaPaCa-2, and primary PancM pancreatic cancer cells to radiation and Gemcitabine
- ATR inhibition by VE-821 leads to inhibition of radiation-induced G2/M arrest in cancer cells
- Inhibits phosphorylation of Chk1 (Ser 345) in PSN-1 and MiaPaCa-2 cells after treatment with Gemcitabine, radiation, or both

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Apexbio Technology LLC VE-821 1232410-49-9 100mg
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
VE-821 (CAS 1232410-49-9) is a selective ATP-competitive inhibitor of ATR (ataxia telangiectasia and Rad3-related) kinase a critical regulator involved in DNA damage response signaling VE-821 exhibits potent ATR inhibition with a Ki value of 13 nM and minimal off-target activity toward other PI3K-related kinases such as ATM DNA-PK mTOR and PI3K In cellular models VE-821 effectively reduces phosphorylation of ATR substrates including Chk1 at Ser345 thereby sensitizing HL-60 leukemia and pancreatic cancer cell lines (e g PSN-1 MiaPaCa-2) to chemotherapy (gemcitabine) and radiation-induced cytotoxicity VE-821 is utilized as a research tool in the study of DNA damage responses and cancer vulnerabilities

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More