Tricarboxylic acids and derivatives
- (2)
- (1)
- (3)
- (1)
- (2)
- (2)
- (2)
- (1)
- (3)
- (1)
- (1)
- (1)
- (1)
- (1)
- (2)
- (1)
- (1)
- (2)
- (3)
- (1)
- (3)
- (2)
- (2)
- (1)
- (2)
Filtered Search Results
Tamoxifen citrate
CAS: 54965-24-1 Molecular Formula: C32H37NO8 Molecular Weight (g/mol): 563.65 MDL Number: MFCD00058321 InChI Key: FQZYTYWMLGAPFJ-OQKDUQJOSA-N Synonym: tamoxifen citrate,istubal,zitazonium,unii-7frv7310n6,i.c.i. 46474 citrate,tamoxifen citrate salt,farmifeno,ginarsan,jenoxifen PubChem CID: 2733525 ChEBI: CHEBI:9397 IUPAC Name: (2-{4-[(1Z)-1,2-diphenylbut-1-en-1-yl]phenoxy}ethyl)dimethylamine; 2-hydroxypropane-1,2,3-tricarboxylic acid SMILES: OC(=O)CC(O)(CC(O)=O)C(O)=O.CC\C(=C(/C1=CC=CC=C1)C1=CC=C(OCCN(C)C)C=C1)C1=CC=CC=C1
| PubChem CID | 2733525 |
|---|---|
| CAS | 54965-24-1 |
| Molecular Weight (g/mol) | 563.65 |
| ChEBI | CHEBI:9397 |
| MDL Number | MFCD00058321 |
| SMILES | OC(=O)CC(O)(CC(O)=O)C(O)=O.CC\C(=C(/C1=CC=CC=C1)C1=CC=C(OCCN(C)C)C=C1)C1=CC=CC=C1 |
| Synonym | tamoxifen citrate,istubal,zitazonium,unii-7frv7310n6,i.c.i. 46474 citrate,tamoxifen citrate salt,farmifeno,ginarsan,jenoxifen |
| IUPAC Name | (2-{4-[(1Z)-1,2-diphenylbut-1-en-1-yl]phenoxy}ethyl)dimethylamine; 2-hydroxypropane-1,2,3-tricarboxylic acid |
| InChI Key | FQZYTYWMLGAPFJ-OQKDUQJOSA-N |
| Molecular Formula | C32H37NO8 |
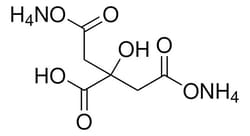
Sigma Aldrich Ammonium citrate dibasic
MilliporeSigma Sigma Organics products encompass a wide range of quality reagents, solvents, catalysts, and building blocks for organic synthesis. From benchtop discovery to process development and scale-up, Sigma Organics solutions are built to meet the needs of synthetic chemists.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
| Percent Purity | ≥98% |
|---|---|
| Linear Formula | HOC(CO2H)(CH2CO2NH4)2 |
| CAS | 3012-65-5 |
| Molecular Weight (g/mol) | 226.18 |
| MDL Number | MFCD00013068 |
| Synonym | Ammonium hydrogencitrate; Citric acid ammonium salt; Diammonium hydrogen citrate |
| RTECS Number | GE7545000 |
| Recommended Storage | Room Temperature |
| Molecular Formula | C6H14N2O7 |
| EINECS Number | 221-146-3 |
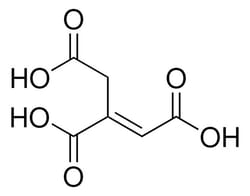
Sigma Aldrich trans-Aconitic acid
MilliporeSigma Sigma Organics products encompass a wide range of quality reagents, solvents, catalysts, and building blocks for organic synthesis. From benchtop discovery to process development and scale-up, Sigma Organics solutions are built to meet the needs of synthetic chemists.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
| Percent Purity | 98% |
|---|---|
| Linear Formula | HO2CCH2C(CO2H)=CHCO2H |
| CAS | 4023-65-8 |
| Molecular Weight (g/mol) | 174.11 |
| MDL Number | MFCD00002721 |
| Synonym | trans-Propene-1,2,3-tricarboxylic acid |
| Recommended Storage | Room Temperature |
| Molecular Formula | C6H6O6 |
| EINECS Number | 223-688-6 |
| Melting Point | 190°C (lit.) |
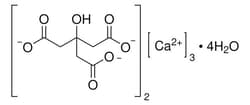
Sigma Aldrich Calcium citrate tetrahydrate
MilliporeSigma Sigma Organics products encompass a wide range of quality reagents, solvents, catalysts, and building blocks for organic synthesis. From benchtop discovery to process development and scale-up, Sigma Organics solutions are built to meet the needs of synthetic chemists.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
| Percent Purity | 99% |
|---|---|
| Linear Formula | [O2CCH2C(OH)(CO2)CH2CO2]2 Ca3 -+ 4H2O |
| CAS | 5785-44-4 |
| Molecular Weight (g/mol) | 570.49 |
| MDL Number | MFCD00150786 |
| Synonym | Tricalcium dicitrate tetrahydrate |
| Recommended Storage | Room Temperature |
| Molecular Formula | C12H10Ca3O14 -+ 4H2O |
| EINECS Number | 212-391-7 |
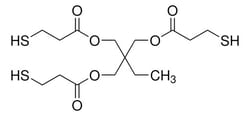
Sigma Aldrich Trimethylolpropane tris(3-mercaptopropionate)
MilliporeSigma Sigma Organics products encompass a wide range of quality reagents, solvents, catalysts, and building blocks for organic synthesis. From benchtop discovery to process development and scale-up, Sigma Organics solutions are built to meet the needs of synthetic chemists.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
| Boiling Point | 220°C (0.3 mmHg) |
|---|---|
| Percent Purity | ≥95.0% |
| Linear Formula | (HSCH2CH2CO2CH2)3 CC2H5 |
| CAS | 33007-83-9 |
| Biological Activity | Respiratory System |
| Molecular Weight (g/mol) | 398.56 |
| MDL Number | MFCD00046842 |
| Refractive Index | n20/D 1.518 (literature) |
| Recommended Storage | Room Temperature |
| Molecular Formula | C15H26O6S3 |
| EINECS Number | 251-336-1 |
| Density | 1.21 g/mL (at 25°C (literature)) |
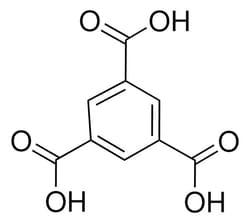
Sigma Aldrich Trimesic acid
MilliporeSigma Sigma Organics products encompass a wide range of quality reagents, solvents, catalysts, and building blocks for organic synthesis. From benchtop discovery to process development and scale-up, Sigma Organics solutions are built to meet the needs of synthetic chemists.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
| Percent Purity | 95% |
|---|---|
| Linear Formula | C6H3(CO2H)3 |
| CAS | 554-95-0 |
| Molecular Weight (g/mol) | 210.14 |
| MDL Number | MFCD00002517 |
| Synonym | BTC; H3BTC; TMA; Benzene-1,3,5-tricarboxylic acid |
| Recommended Storage | Room Temperature |
| Molecular Formula | C9H6O6 |
| EINECS Number | 209-077-7 |
| Melting Point | >300°C (lit.) |
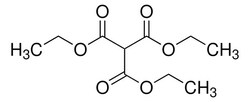
Sigma Aldrich Triethyl methanetricarboxylate
MilliporeSigma Sigma Organics products encompass a wide range of quality reagents, solvents, catalysts, and building blocks for organic synthesis. From benchtop discovery to process development and scale-up, Sigma Organics solutions are built to meet the needs of synthetic chemists.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
| CAS | 6279-86-3 |
|---|
Medchemexpress LLC Azido-PEG2-CH2COOH | 882518-90-3 | 99.03% | 189.17 | 1 ML
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Azido-PEG2-CH2COOH is a PEG-based PROTAC linker used in the synthesis of PROTACs. It is a click chemistry reagent containing an Azide group that can undergo copper-catalyzed azide-alkyne cycloaddition reaction (CuAAc) with molecules containing Alkyne groups. It can also undergo strain-promoted alkyne-azide cycloaddition (SPAAC) reactions with molecules containing DBCO or BCN groups. PROTACs use the intracellular ubiquitin-proteasome system to degrade target proteins.
- PEG-based PROTAC linker.
- Click chemistry reagent.
- Contains an Azide group for CuAAc reaction with Alkyne groups.
- Can undergo SPAAC reactions with DBCO or BCN groups.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC Azido-PEG6-CH2COOH | 880129-82-8 | ≥98.0% | 365.38 | 1 G
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Azido-PEG6-CH2COOH is a PEG-based PROTAC linker used in the synthesis of PROTACs. It is a click chemistry reagent, featuring an azide group for copper-catalyzed azide-alkyne cycloaddition (CuAAc) reactions with alkyne-containing molecules. It also facilitates strain-promoted alkyne-azide cycloaddition (SPAAC) reactions with DBCO or BCN groups.
- Functions as a PEG-based PROTAC linker
- Used in the synthesis of PROTACs
- Acts as a click chemistry reagent
- Enables copper-catalyzed azide-alkyne cycloaddition (CuAAc) reactions
- Supports strain-promoted alkyne-azide cycloaddition (SPAAC) reactions
- Integral to PROTACs that selectively degrade target proteins

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC Acetic acid, 2-[2-(2-azidoethoxy)ethoxy]- | 882518-90-3 | 99.0% | 189.17 | 1 MG
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Azido-PEG2-CH2COOH is a PEG-based PROTAC linker utilized in the synthesis of PROTACs. It functions as a click chemistry reagent due to its Azide group, enabling it to undergo copper-catalyzed azide-alkyne cycloaddition reactions (CuAAc) with alkyne-containing molecules. Additionally, it can participate in strain-promoted alkyne-azide cycloaddition (SPAAC) reactions with molecules possessing DBCO or BCN groups.
- PEG-based PROTAC linker
- Click chemistry reagent
- Contains an Azide group
- Undergoes copper-catalyzed azide-alkyne cycloaddition reaction (CuAAc)
- Undergoes strain-promoted alkyne-azide cycloaddition (SPAAC) reactions
- Can be used in the synthesis of PROTACs
- PROTACs utilize the intracellular ubiquitin-proteasome system to degrade target proteins

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC Acetic acid, 2-[2-(2-azidoethoxy)ethoxy]- | 882518-90-3 | 99.03% | 189.17 | 50 MG
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Azido-PEG2-CH2COOH is a PEG-based PROTAC linker that can be used in the synthesis of PROTACs. It is also a click chemistry reagent, containing an Azide group that can undergo copper-catalyzed azide-alkyne cycloaddition reaction (CuAAc) with molecules containing Alkyne groups. It can also undergo strain-promoted alkyne-azide cycloaddition (SPAAC) reactions with molecules containing DBCO or BCN groups. This product is for research use only.
- Peg-based PROTAC linker
- Click chemistry reagent
- Contains an Azide group for CuAAc and SPAAC reactions

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC Azido-PEG6-CH2COOH | 880129-82-8 | ≥98.0% | 365.38 | 5 G
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Azido-PEG6-CH2COOH is a PEG-based PROTAC linker used in the synthesis of PROTACs. It functions as a click chemistry reagent, enabling copper-catalyzed azide-alkyne cycloaddition (CuAAc) with alkyne-containing molecules and strain-promoted alkyne-azide cycloaddition (SPAAC) reactions with molecules containing DBCO or BCN groups.
- PEG-based PROTAC linker
- Suitable for synthesizing PROTACs
- Click chemistry reagent due to azide group
- Enables CuAAc reactions
- Participates in SPAAC reactions with DBCO or BCN groups

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC Acetic acid, 2-[2-(2-azidoethoxy)ethoxy]- | 882518-90-3 | 99.0% | 189.17 | 100 MG
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Azido-PEG2-CH2COOH is a PEG-based PROTAC linker utilized in the synthesis of PROTACs. It functions as a click chemistry reagent, containing an Azide group that can participate in copper-catalyzed azide-alkyne cycloaddition (CuAAc) reactions with alkyne-containing molecules. It can also engage in strain-promoted alkyne-azide cycloaddition (SPAAC) reactions with molecules possessing DBCO or BCN groups.
- Functions as a click chemistry reagent
- Contains an azide group
- Participates in copper-catalyzed azide-alkyne cycloaddition (CuAAc) reactions
- Engages in strain-promoted alkyne-azide cycloaddition (SPAAC) reactions

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC Acetic acid, 2-[2-(2-azidoethoxy)ethoxy]- | 882518-90-3 | 99.03% | 189.17 | 10 MG
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Azido-PEG2-CH2COOH is a PEG-based PROTAC linker designed for use in the synthesis of PROTACs. It also functions as a click chemistry reagent, featuring an Azide group that can participate in copper-catalyzed azide-alkyne cycloaddition reactions (CuAAc) with molecules containing Alkyne groups. Additionally, it can undergo strain-promoted alkyne-azide cycloaddition (SPAAC) reactions with molecules containing DBCO or BCN groups.
- Can be used in the synthesis of PROTACs.
- Functions as a click chemistry reagent.
- Azide group enables copper-catalyzed azide-alkyne cycloaddition.
- Can undergo strain-promoted alkyne-azide cycloaddition with DBCO or BCN groups.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More
Medchemexpress LLC Fmoc-NH-PEG1-CH2COOH | 260367-12-2 | MFCD06796876 | 99.9% | 341.36 g·mol⁻¹ | C19H19NO5 | 25 G
 Small and Specialty Supplier Partner
Small and Specialty Supplier Partner
Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More

Small and/or specialty supplier based on Federal laws and SBA requirements.
Learn More
Fmoc-NH-PEG1-CH2COOH is an Fmoc-protected PEG1 linker bearing a terminal carboxylic acid used as a cleavable linker in antibody-drug conjugate (ADC) synthesis and as a PEG-based PROTAC linker in conjugation chemistry. It has CAS number 260367-12-2, molecular formula C19H19NO5, and a molecular weight of 341.36 g·mol⁻¹.
- Cleavable ADC linker for antibody-drug conjugation.
- Suitable as a PEG-based PROTAC linker.
- Fmoc-protected amine enables standard peptide and solid-phase chemistries.
- High reported purity suitable for synthetic applications.
- Available in multiple pack sizes including 25 G for preparative work.

Non-distribution item offered as a customer accommodation; additional freight charges may apply.
Learn More